
CASE NOTES
Swine Pox Infection in Non-Commercial Pig Herd NW NSW
Justine McNally BVSc., Moree District Veterinarian
Posted Flock & Herd August 2022
Introduction
Swine pox (SwVP) is not a common diagnosis in North West (NW) New South Wales (NSW), largely because there are few domestic pig operations of any size in our area and because the disease has been effectively controlled within the commercial industry. The case investigated was a typical scenario that non-pig industry veterinarians may be faced with when called to investigate a potential swine pox outbreak in a non-commercial pig herd.
Case history
A farmer in the Moree district called me in late January as five piglets from his most recent litter presented with multiple, round, raised lesions over their entire bodies. These piglets had been farrowed from a gilt on 01/01/2022. The farmer had one boar, one 2½ years old sow, two 10-12 months old sows and seven growers approximately three months of age, as well as the five affected piglets.
The pigs were kept in a set of old cattle yards, which were surrounded by some grass and herbage and cultivated paddocks. There was an underdeveloped area to the north-east of the yards that ran back into a tree line. The property was enclosed by exclusion fencing (Weston Fence) on three of the four boundaries. The owner had not seen any evidence of feral pigs in the vicinity over the last couple of years, but it must be noted that his domestic pigs had only been resident for a few months and feral pigs are prevalent in this watercourse country.
The piglets were the only animals within this herd that were displaying the skin lesions. All the older pigs appeared to be healthy and content.
Before proceeding to the property, I consulted Swine Health and Production - Vol 3 (2) to give some direction with my investigation.
Clinical examination
The piglets presented with multifocal, raised, well-circumscribed lesions approximately 0.7-1.0cm in diameter. The surface of the lesions was crusted, with the centre of the lesion either slightly depressed or raised, but all appeared to have a red-to-purple ring surrounding the central part of the lesion creating a collarette.
Lesions were seen on all epidermal surfaces of the body - ventral and dorsal aspects of the torso, legs, ears, face and snout. Both the hairy and non-hairy areas seemed to be similarly affected.
There were no lesions present on the mucosal surfaces of the oral cavity.
Body condition score of the piglets was 2/5 and the owner commented that they were not as fat, even at birth, as the previous litter.



Images 1-3. A series of photos taken of affected piglet, 20th January 2022 (J McNally)
SAMPLE COLLECTION
To establish a diagnosis an affected piglet was caught and examined.
There were multiple lesions across the entire body, so to visualise them more clearly an area on the right side of the body was cleaned with povidine iodine scrub and shaved. A chlorhexidine/alcohol solution was then used to wipe the area, further cleaning gross contamination from the skin and lesions. After this preparation it was noted that some of the lesions were pustules rather than crusts. Some were centrally raised and yellow to light brown in colour. The tissue surrounding this central area was light pink and it was surrounded by a hyperaemic collar. For some lesions the central area was depressed, and the peripheral tissue was thickened.
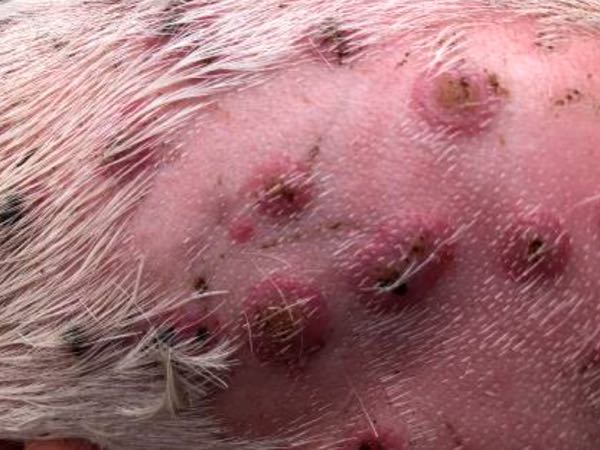
Image 4. Shaved and aseptically prepared site, (J McNally 20th January 2022)
One lesion was scrubbed and aseptically prepared. A biopsy was taken from this area and included both affected and normal tissue. This tissue was placed in 10% formalin.
A skin scraping was taken of an unprepared lesion as well as a tape impression from another affected, unprepared area.
A crust was removed and placed in 10% formalin using sterile forceps and a swab was then taken from the area beneath the crust for bacterial culture. Another crust was removed and placed in phosphate-buffered saline with glucose (PBSG).
Blood was also collected.
Differential diagnosis
Speculative diagnosis at this stage was for a pox-like disease but due to my lack of experience with pig medicine my list of differentials was extensive. The list included:
- Swine pox (SwVP)
- Fungal infection - ringworm
- Pityriasis rosea
- Erysipelas
- Parvovirus
- Greasy Pig disease (Staphylococcus hyicus)
- Parasitic skin diseases
- Allergic skin lesions
- Porcine dermatitis and nephropathy syndrome
- Foot-and-mouth Disease
- Vesicular exanthema
- Vesicular stomatitis
- Swine vesicular disease
On history and clinical examination, coupled with lesion appearance, pattern and distribution, I was able to rule out the major emergency animal diseases (EADs). This exclusion was based on the fact lesions were not evident on the mucosal surfaces and coronary bands and the clinical signs only affected the young piglets despite pigs of all ages being in contact with each other. Also, despite the piglets being ill-thrifty they were not depressed or lethargic.
The differential diagnoses highest on my list at point of submission were:
- Swine pox
- Fungal infection - Trichophyton verrucosum (ringworm)
- Pityriasis rosea
- Erysipelas
- Staphylococcus hyicus - Greasy Pig
Laboratory testing
Samples were sent the NSW State Animal Laboratory at the Elizabeth Macarthur Agricultural Institute (EMAI) at Menangle.
The tests deemed appropriate to conduct by the pathologist were:
- Histology
- Bacterial culture
- Microscopic examination of the tape impressions and skin scrapings
- Fungal culture
- Electron microscopy (EM) -ve stain.
Laboratory results
There was no growth on the fungal culture and no evidence of ectoparasites.
The histopathology and EM examinations confirmed the involvement of poxviral infection.
On histopathology, changes consistent with poxviral infection were present, including, 'epithelial cells are often enlarged with clear cytoplasm (ballooning degeneration) and contain occasional eosinophilic cytoplasmic inclusion bodies' (A. Jordan, laboratory report). Histopathology and EM were consistent with pox virus infection, presumed swine pox, especially when taken in conjunction with signalment, clinical findings and history.

Change from normal epidermis to hyperplastic epidermis (A. Jordan)

Intracytoplasmic inclusions (A. Jordan)
On EM, poxvirus particles were detected, which confirmed the diagnosis of swinepox.
Staphylococcus hyicus was isolated but this bacterial species was viewed as an opportunistic secondary invader.
Discussion
Swine pox belongs to the subfamily Chorodopoxvirnae within the Poxviridae family and is the only member of the genus Suispoxvirus. It was first reported in Europe in 1842 and it occurs sporadically throughout the world. Only pigs are susceptible to this poxvirus. It is not a zoonoses as SwVP is highly host specific.
Transmission of the virus between pigs appears to commonly occur by the bite of the pig louse (Haematopinus suis). The prevalence of lice is influenced by general sanitation, geographical influences and contact with other pig populations. It is thought that mosquitoes and flies may also be vectors. Horizontal transmission between pigs may also occur via direct contact of oral and nasal secretions and desquamated scabs into skin abrasions. Occasionally there are sporadic cases of congenital swinepox due to vertical transmission (transplancental).
Pathogenesis
SwVP enters the host through a break in the skin and replicates in the cytoplasm of the keratinocytes of the stratum spinosum1. So, injury of the skin is required for the infection to start5. The pig louse (Haematopinus suis) is commonly implicated in outbreaks of SwVP as they act as mechanical vector for the virus1,2,3,4,5,6; but direct contact3,4,5,6 of oral or nasal secretions, or desquamated scabs from a carrier/infected pig to a susceptible individual can also lead to expression of the disease1. From as early as three days post infection, SwVP can be isolated from skin lesion material1. It is not known how the virus spreads from the initial site of replication to other areas of the body1.

Pig louse (J McNally)
Clinical Signs
Pigs of all ages can be infected with SwVP1,5 with morbidity in a herd potentially reaching 100%3. But it is piglets <4 months of age that are most severely affected1,2,3,5,6. Despite the morbidity rate being so high, the mortality rate is low1,2. In adults, the clinical signs are mild and self-limiting1 or do not appear at all due to acquired immunity from a prior infection5. In piglets the disease presents as a typical pox disease with the development of cutaneous lesions that progress through classical stages of macule, papule, vesicle, pustule to scab/crust and then resolution1,3,4,5.
Lesions are commonly found in areas frequented by the louse, such as the ventral abdomen, inguinal areas and ears1,3,6. The lesions then progress across the entire body to incorporate the flanks, torso, legs and face1,4. Occasionally lesions may be seen around the bulbs of the heels, along coronet bands, lips, tongue and oral mucosa so care must be taken to consider EADs in one's differential diagnosis list1,5.
The incubation period is 3-14 days in length, at which point the lesions begin to develop1,5. During this initial phase, the macular stage, the lesions are pale, flat and round but within 48 hours develop into a raised papule approximately 1cm in diameter. The lesions then become vesicles, which may not be observed1,5 as they progress rapidly to pustules and by about one week form a crust. These crusts remain for approximately three weeks5. Once they have desquamated they leave a small, white macule1,5.

Crusted lesions (Lesion as seen on 9th February 2022 - J. McNally)

Desquamating lesion (Lesion as seen on 9th February 2022 - J. McNally)

White Macule (Lesion as seen on 9th February 2022 - J. McNally)
Conclusion
Swine Pox is not common now as large-scale, modern pig producers enforce tight biosecurity measures and high levels of hygiene, and there are fewer small-scale piggeries run as part of a mixed farming operations.
With the increase in pig ownership, on a backyard or hobby level, SwVP is something that should be considered when examining piglets with skin lesions, especially in areas where feral pig populations are close to pig enclosures as feral pigs can carry both lice and the virus.
This case proved to be both interesting and a wonderful learning experience as most general veterinarians do not have the opportunity to investigate pig diseases. An understanding of this disease, including both its expression and transmission, as well as the differential diagnoses, is important for all veterinarians not familiar with pig diseases or investigating pig diseases. This understanding is becoming increasingly important as pig ownership on a small scale appears to be increasing and vigilance for EADs is always at the forefront of Australia's livestock surveillance programs.
BIBLIOGRAPHY
- Zimmerman J.J. et al. Diseases of Swine, 10th ed. 2019. John Wiley & Sons, Inc., pp 709-713
- Doster A.R., Swine Health and Production, 1995, 3(6): 256-260
- Mech P. et al. Virus Disease, 2018, 29(3): 395-399
- Thibault S. et al. Swine Health and Production, 1998, 6(6): 276-278
- Kasza, L. Diseases of Swine, 4th ed., 1975, Editors Dunne, H.W. & Leman A.D., 2(10): 273-285
- Kaiser F.K. et al. Viruses, 2021, 13, 2038: 1-15
Acknowledgements
Many thanks to Anne Jordan, Pathologist EMAI, for her assistance with this case and the great photos of the histological changes.