
CASE NOTES
Photosensitisation and cholangiohepatopathy (Dikoor) in lambs grazing a forage brassica crop contaminated with Panicum gilvum
Bruce Watt, (Tablelands LHPA Bathurst) and Belinda Hackney (formerly of NSW DPI Bathurst) and Erika Bunker (State Veterinary Diagnostic Laboratory, Menangle)
Posted Flock & Herd July 2013
Introduction
The brassicas are a genus of plants that contain some of the world's most important oil-seed, forage, vegetable and weed species. However, when grazed, they can cause several livestock health problems including goitre, hypothyroidism, polioencephalomalacia, enteritis, acute pulmonary emphysema and interstitial pneumonia, haemolytic anaemia and nitrate poisoning. Primary photosensitisation is a well-known adverse consequence of grazing brassicas (Radostits et al. 2007). However, brassicas are not reported as causing cholangiopathy or hepatopathy. This is a report of a case of crystal-associated cholangiohepatopathy in lambs grazing Winfred forage brassica (a cross between a turnip and kale). However, other plants including Panicum gilvum, a known cause of cholangiohepatopathy (Lancaster 1999), contaminated the crop. In addition, there was evidence that the Panicum had been heavily grazed whereas the brassica had not. It is therefore presumed that the Panicum was the primary cause of this outbreak.
History
On 4 December 2012, the owner of 520 August 2012 drop second cross lambs found six dead and noticed 30-40 with swollen ears and faces. The lambs had been weaned onto a crop of Winfred forage brassica plus Tonic plantain (Plantago lanceolata, a deep-rooted forage herb) on the 26 November 2012. The 26 ha crop had been sown into a cultivated paddock eleven weeks previously. The crop was contaminated with a range of other plants including a grass that was subsequently identified as Panicum gilvum. Despite moving the lambs to a shaded pasture and a clover hay aftermath paddock, 42 lambs died or were euthanased.

Figure 1. The crop of Winfred forage brassica
Clinical and necropsy findings
Three lambs were examined and one was necropsied on the 6 December 2012. The remainder of the mob was inspected in the new, shaded paddock. All lambs examined had marked photosensitisation with swollen ears and faces. All lambs were dull and lethargic. Case 1 had yellow mucous membranes and pronounced swelling of face and ears while cases 2 and 3 were not noticeably jaundiced. Case 1 was necropsied revealing swollen, yellow, gelatinous sub-mandibular tissues. The liver was dark red but of normal size with sharp edges. The gastrointestinal contents were dry and scant.

Figure 2. Affected lambs, distressed with swollen faces and ears, seeking shade
The property was revisited on 12 December 2012 as the owner elected to euthanase the badly affected lambs because they had deteriorated and appeared to be suffering. Six of these lambs, weighing between 25 and 35 kg, were necropsied. All had necrosis of the skin around the eyes and frequently on the lips and all were dull, weak and dehydrated. On necropsy, all were moderately to markedly jaundiced. Five of the six (cases 5-9) had slightly to moderately enlarged livers that felt turgid and were olive in colour. Case 7 had haemorrhages in the wall of the gall bladder. Five of the six (5-9) had kidneys that appeared swollen, friable and had dark grey mottling over the external surface. Cases 7 and 9 had evidence of pneumonia in the apical lung lobes.

Figure 3. Necrosis of skin around the eyes, ears and lips (Case 4, 12 December 2012)
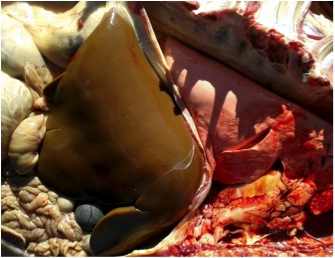
Figure 4. Enlarged olive coloured liver. (Case 5, 12 December 2012)

Figure 5. Swollen mottled kidney (Case 7, 12 December 2012)
Clinical pathology
There was no evidence of anaemia on bloods collected from cases 1 to 9. Some lambs had an increased PCV and protein levels indicating dehydration and most had a neutrophilia.
Most lambs had markedly increased levels of GGT and conjugated bilirubin, indicating bile stasis. There was increase in AST without concurrent increase in CK (suggesting hepatocellular damage rather than muscle damage). Cases 1-3 had increased GLDH levels. Blood copper levels from cases 1-3, collected on 6 December 2012, were elevated, ranging from 23.9 to 28.1 mmol/L (normal range 7.5 to 16.0 mmol/L). Blood copper levels from cases 4-9 (collected on the 12 December) were further elevated, ranging from 33.5 to 63.6 mmol/L.
Histopathology
Liver and kidney samples from cases 1 and 4 to 9 were examined. In addition, the gall bladder of case 7 and lung from cases 7 and 9 that showed gross lesions were also examined. All lambs had a severe subacute lympho-plasmacytic cholangiohepatitis with crystal clefts and varying degrees of Kupffer cell hyperplasia, bile stasis and single cell necrosis. Case 1 also had a multifocal necro-suppurative hepatitis. There was a tubulonephrosis with crystal clefts and lympho-plasmacytic interstitial nephritis in some kidneys. Acute suppurative bronchopneumonia was diagnosed on histopathology of lung samples with gross lesions.
Paddock inspection
The paddock was inspected (by BH) on 12 December 2012. At the time of inspection, it was composed of 73% Brassica, 9% ryegrass (Lolium spp), 12% Panicum, 3% soft brome (Bromus mollus), 2% vulpia (Vulpia bromoides) and 1% mixed broad leaf (predominately annual clovers - Trifolium ceruum or T. striatum).
The Winfred forage brassica was on average 7.5 cm high, although this varied across the paddock and ranged from 4-20 cm. The Panicum and the ryegrass had both been grazed heavily and consistently across the paddock to approximately 3 cm in height. The brassica leaves were intact indicating minimal grazing within the past 10 days and approximately 30% of the paddock had brassica plants >15 cm high suggesting that they had not been grazed at all. This variability in the height of the crop was probably a consequence of paddock differences. For example, the higher areas of the paddock where sheep had presumably camped had the tallest crop whilst the lower areas, such as gullies had the shortest, possibly reflecting increased utilisation and/or poor drainage.
Plant identification
Samples of the grasses requiring identification were collected from the paddock on 12 December 2012 and grown to maturity in pots at the Bathurst Agricultural Research and Advisory Station. Samples were then submitted to the National Herbarium of NSW for identification. They were identified as Panicum gilvum.
Subsequent management
The owner returned the surviving lambs to the forage brassica crop in late December 2012 after the crop responded to storm rains. The lambs grew well with no further health problems.
Discussion
All lambs necropsied had a crystal-associated cholangiohepatopathy consistent with intoxication by steroidal saponins. The photosensitisation and hepatobiliary disease that characterise this disorder resembles geeldikkop, which is known to occur when sheep graze Tribulus terrestris plus an unknown secondary factor, possibly a mycotoxin. Geeldikkop has been described as 'the last of the great endemic diseases of South Africa,' with outbreaks affecting more than half a million sheep (Kellerman et al. 1991). Geeldikkop also causes sheep losses in Australia (Glastonbury et al. 1984; Bourke CA 1983; Jacob RH and Peet RL 1987).
The panics, most notably Panicum effusum (hairy panic), Panicum capillare (witchgrass) and Panicum miliaceum (French millet) together with a second unknown factor have also caused crystal-associated cholangiohepatopathy in sheep in Australia and elsewhere (Hurst E 1942, Everist SL 1974, McBarron 1976, Kellerman et al. 1991). In South Africa, this disease is referred to as dikoor (Kellerman et al. 1991). In Victoria, hepatogenous photosensitisation occurred in lambs that grazed paddocks where Panicum schinzii was identified (Button C et al. 1987; Lancaster MJ et al. 1991). Lancaster (1999) stated that Panicum gilvum, previously misidentified as P. schinzii, was the plant most commonly associated with this syndrome in northeastern Victoria.
While it was initially suspected that this was an extreme form of photosensitisation with cholangiohepatopathy caused by the Winfred forage brassica, the subsequent identification of Panicum gilvum, a known source of steroidal saponins, obscures the role of the brassica. While the brassica may have exacerbated the photosensitisation, it is presumed that the panic was the primary source of toxin.
Acknowledgements
We would like to acknowledge the owner of these lambs whose willing co-operation enabled us to investigate this case in detail. We would also like to acknowledge Adrian Lynch (NSW DPI) who grew the panic and submitted it for identification and the National Herbarium of NSW for identifying Panicum gilvum.
References
- Bourke CA (1983) Hepatopathy in sheep associated with Tribulus terrestris. Australian Veterinary Journal 60(6):189
- Button C, Paynter DI, Shiel MJ, Colson AR, Paterson PJ and Lyford RL (1987) Crystal-associated cholangiohepatopathy and photosensitisation in lambs Australian Veterinary Journal 64(6):176-187
- Everist SL (1974) Poisonous Plants of Australia pp 331-334
- Glastonbury JRW, Doughty SJ, Whittaker SJ and Sergeant E (1984) A syndrome of hepatogenous photosensitisation, resembling geeldikkop, in sheep grazing Tribulus terrestris. Australian Veterinary Journal 61(10):314-316
- Hurst E (1942) The Poison Plants of New South Wales pp 19-20
- Jacob RH and Peet RL (1987) Poisoning of sheep and goats by Tribulus terrestris (caltrop) Australian Veterinary Journal 64(9):288-289
- Kellerman TS, Erasmus GL, Coetzer JAW, Brown JMM and Maartens BP (1991) Photosensitivity in South Africa. VI. The Experimental Induction of Geeldikkop in sheep with Crude Steroidal Saponins from Tribulus terrestris Onderstepoort Journal of Veterinary Research 58:47-53
- Lancaster MJ (1999) Steroidal sapogenin intoxication of cavies and rats: comparison with Panicum gilvum intoxication of cavies and sheep Proceedings Australian Society for Veterinary Pathology Conference Melbourne 1999 p 24
- Lancaster MJ, Vit I and Lyford RL (1991) Analysis of bile crystals from sheep grazing Panicum schinzii (sweet grass) Australian Veterinary Journal 68(8):281
- McBarron EJ (1976) Medical and Veterinary Aspects of Plant Poisons in New South Wales
- Radostits OM, Gay CC, Hinchcliff KW and Constable PD (2007) Veterinary Medicine, 10th Edition