
ARCHIVE FILE
This article was published in 1987
See the original document
Porcine Parvovirus (PPV) Outbreaks in the North West of New South Wales
A Discussion on the Field Epidemiology
J A MACFARLANE BvSC, Veterinary Inspector ARMIDALE (Ex NARRABRI)
Introduction
The field data in this paper was gathered while I was the Veterinary Inspector at Narrabri PP Board, and consequently is restricted to that area. However in speaking with veterinarians in other parts of Australia I have no reason to believe it is any different anywhere else.
There are no major piggeries in the area, but the general distribution of herd sizes is similar to the State average, with the three largest being 250 - 300 sows each.
The record keeping systems are no better than average and only these on the AIMS (Reg IM) Computer Recording System really allow scientific evaluation, hence some of the figures regarding parvovirus outbreaks are educated guesses.
Rather than just presenting a number of cases, some of dubious scientific accuracy: I will give you my idea of the epidemiology of the disease. This was developed from various literature reports and substantiated by local data.
Pathogenesis
PPV causes reproductive disorders in pregnant susceptible (i.e. antibody negative) gilts and sows. The disease pattern has been well researched and a summary by Wrathall (1984) is given below:
Figure 1
Manifestations of PPV disease in relation to stage of pregnancy when infections occurs.
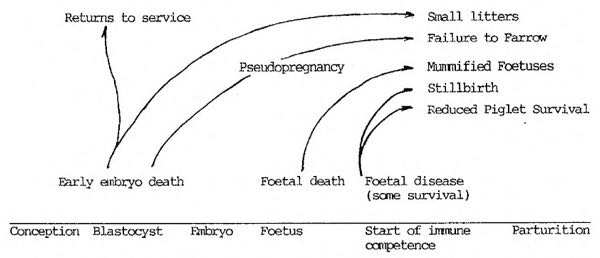
In practical terms the earliest parameter with which the progress of the disease can be measured is the percentage of mummified foetuses - this correlates with the fall in the number born alive.
A recent recorded outbreak in Narrabri amongst a large number of introduced susceptible gilts gave the following figures.
Table 1 (sic)
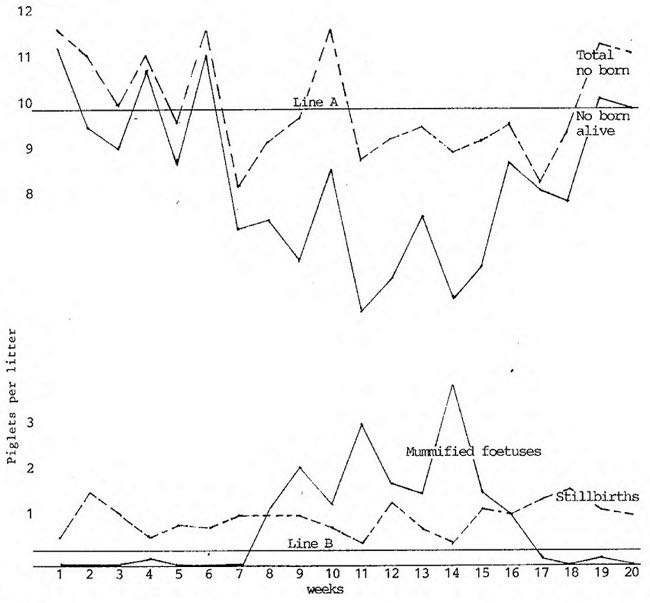
Line A
Acceptable tolerance level below which investigation or action should occur for total number born alive.
Line B
Acceptable tolerance level above which investigation should occur for mummified foetuses.
Naturally this situation only occurs in a fully susceptible population. The extent of any problem is based on the number of susceptible animals at any one time and it is this area that I am hoping to clarify by a discussion of the epidemiology of the disease.
Epidemiology
Herd Prevalence
As PPV reproductive failure only occurs in seronegative pregnant gilts and sows, the serological status of a herd will indicate the potential for reproductive failure in the event of the herd outbreak.
Earlier work suggested that PPV was endemic in up to 90% of Australian herds (Johnson and Nunn 1979) and that 90 - 100% of adult stock on these farms were serologically positive to PPV (Johnson et al. 1976). Earlier work also suggested that the introduction of virus to any herd free of PPV would result in nearly 100% infection in the herd within 3 months (Donaldson-Wood et al. 1977).
A recent Australian report (Johnson 1984) has re-defined the position. This report divides herds into three categories based on the serological status of sows and gilts; and hence numbers at risk to infection. These are:
1. In an estimated 60% of herds virtually all sows and 85% of gilts will have acquired an active immunity prior to their first service.
2. 35% of herds contain a variable percentage (15 - 60%) of adult sows and gilts which have not developed active immunity and are therefore susceptible to PPV reproductive failure.
3. 5% of herds have no PPV on the premises and all breeding stock are therefore at risk. These are usually closed, isolated herds with high standards of hygiene and disease control.
Parvovirus Prevalence
All herds fit into one of 3 categories of parvovirus prevalence, i.e.:
Table 2
| 1 | 2 | 3 | |
|---|---|---|---|
| Percentage of herds in each category | 60% | 35% | 5% |
| Viral presence | Widespread | Variable | Absent |
| Sows seropositive | 100% | 15 - 60% | 0 |
| Sows and gilts susceptible (seronegative) | small number gilts (>15%) | 40 - 85% | 100% |
| Disease risk status | Low risk | Mod risk | High risk |
Surveys in other countries that can be interpreted in this manner have given similar findings. Sorenson (1982) reported that in excess of 90% of sows were positive in 57% of 33 herds, with a variable number in the remaining herds. Muirhead et al. (1983) reported over 90% infection rate in 61% of 23 herds.
Disease Outbreak Patterns
In each of these categories a different clinical disease will occur. These have been described by Stein and Lenan (1982) and Muirhead et al. (1983), and basically reflect the disease pattern that occurs when the various susceptible animals within each group become infected. These are:
1. An acute outbreak of reproductive failure will occur in category (3) above if PPV infection occurs. It will last a relatively short time but up to two thirds of pigs farrowed during that time will show the various clinical manifestations of PPV reproductive failure (ie Donaldson-Wood et al. 1977).
2. Acute outbreaks in endemically infected herds will occur in category (2) when a significant population of older animals became susceptible and at risk to reproductive failure. Muirhead et al.. (1983) state that in their experience overt disease in endemically infected herds has become more prevalent.
3. Continual foetal loss in susceptible gilts occurs in herds in category (1).
With regard to this latter group, those gilts which have not been actively infected prior to their first service are susceptible to reproductive failure. Maternal antibody in gilts prevents the development of active immunity following infection (Jahnson et al. 1976), therefore active infection must occur between the loss of maternal antibody and the first service.
Decline of Maternal Antibody to PPV in Gilts
Table 3 (sic)
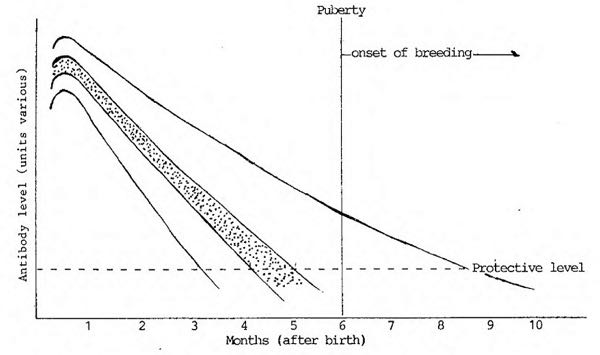
At one time it was thought (Johnson et al. 1976) that maternal antibody was still present at 9 months which would mean that gilts would became susceptible at a time when reproductive disease would occur if active vital infection occurred (Line A).
More recent studies have re-evaluated this and considered that the antibody had disappeared by 4-5 months (ie hatched area in Table 3). Too and Love (1985) reported that maternal antibody had disappeared by 22 weeks.
Varnier et al. (1984) reported that it had disappeared by 14 weeks (Line B). This means gilts have a reasonable period of time to become actively infected and immune before breeding occurs.
Practical Considerations
Experience in Narrabri has been that acute 'outbreaks' of the disease occur every so often (3-10 years) in herds in which the virus is endemic. That is overt disease is occurring in large numbers sporadically in endemically infected herds, rather than constantly in gilts. Muirhead (1983) reported a similar finding in England, as have Vannier et al. (1982) in France and Pointon et al. (1982) in Australia.
The implication is that in between outbreaks the virus is inactive and no outbreaks occur. As a result seronegative (i.e. susceptible) gilts gradually replace older immune sows allowing the build-up of a new susceptible population. Wrathall (1984) has described a characteristic pattern by analysis of PPV serology results from sows of different parities over a number of years as shown in Table 4.
Field Epidemiological Patterns - Serological Variation within a Herd
Table 4
| Year | Sow Parity | Comments | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | ||
| 1 | - | + | + | + | + | + | + | Results immediately after herd outbreak |
| 2 | - | - | - | + | + | + | + | Virus inactive - susceptible gilts |
| 3 | - | - | - | - | - | + | + | Replace older sows |
| 4 | + | + | + | + | + | + | + | New outbreak |
In a Narrabri herd 34 sows were tested. All sows over 5 parities were positive or doubtful (24) and 10 younger sows were all negative. The lab report noted this was a high risk herd and the owners subsequently reported a series of mummified foetuses born to their gilts. These were not quantified.
In another herd in which a sample of 10 ex 10 were positive 6 years ago, none ex 10 were positive five year[s] later and no disease had been seen. This herd is another high risk herd.
In another herd that had recently reported a series of reproductive problems, 20 ex 22 were positive, with 2 younger gilts negative.
My current evaluation of the situation is:
| Hard (sic) Type of Infection | Endemic | Endemic | Endemic | Absent |
|---|---|---|---|---|
| % herds in Narrabri PP Board | 0 | 50 | 50 | 0 |
| In Australia | >5% | 45 | 45 | >5% |
| Presence of virus | widespread & constant | widespread & recent | present but inactive | not present |
| Type of infection | constant & sporadic acute outbreak | Nil | ||
| Sows infected (ie sero positive) | 95 - 100% | 80 - 90% | 70 - 80% | 0 |
| Sows and gilts susceptible | 15% gilts | few ————-> a lot (ie increasing) | 100 | |
| Comment | Confined to large 'dirty' herds where purposeful contamination is practiced. | Sporadic acute outbreaks increasing risk with time. | Closed piggeries only. | |
Prevention
Vaccines
Vaccines are currently available in most (if not all) countries of the World except Australia. Quarantine forbids the introduction of overseas produced vaccines into Australia. CSL are currently developing a local vaccine.
Most overseas commercial vaccines are usually inactivated, i.e. pigs require constant vaccination, similar to the current Erysipelas or Leptospirosis vaccinations. Overseas PPV vaccine is combined with Leptospirosis vaccine.
Although vaccination is not cheap, the cost has been found to be covered by one outbreak, so where available it is recommended routinely on economic grounds (Alexander et al. 1985).
Other Measures
The practice of feeding mummified foetuses does not generally work because it is only during an outbreak these will be due to (and therefore contain) virus and at this time all sows and gilts will be contaminated anyway. Besides it represents an unnecessary risk of spreading other infectious diseases.
The practice of running gilts with baconers during the supposed 'active' phase,i.e. after maternal antibody has gone (20-25 weeks) only seems to work in the large 'dirty' piggeries.
Studies in France (Vannier et al. 1984) and in England (Muirhead et al. 1983) failed to demonstrate the presence of the virus in baconers age in smaller units, or for this practice to assist in sero conversion.
It appears that the virus in smaller piggeries is only active during periods when it is apparent by outbreaks in susceptible pregnant gilts and sows.
Acknowledgements
The assistance of the personnel at Central Veterinary Laboratory, Glenfield in doing all the serological work, is gratefully acknowledged.
This work was partially financed by the MC Garvie Smith Institute and that assistance is gratefully acknowledged.
References
Alexander AM (1985): 'Parvovirus Infection in Pigs'. New Zealand Veterinary Journal 33:93
Donaldson-Wood CR, Joo HS and Johnson RH (1977): 'The effect on reproductive performance of porcine parvovirus infection in a susceptible pig herd' Vet. Rec. 100:237-239
Johnson RH (1984): 'Is there a need for a porcine parvovirus vaccine'. In Proceedings NSW AVA Conference Sydney 20:43. AVA Artarmon NSW 2064
Johnson RH and Nunn MJ (1979): 'Vaccine control of porcine parvovirus reproductive failure'. In Australian Advances in Veterinary Science 1979 (edited by MG Cooper). Australian Veterinary Association, pages 41-42. Artarmon NSW 2064
Johnson RH, Donaldson-Wood CR, Joo HS and Allender U (1976): 'Observations on the epidemiology of porcine parvovirus'. Australian Veterinary Journal 52:80-84
Muirhead MR, Kingston NG and Cook AJC (1983): 'Porcine parvovirus infection - field observations and costs of the disease'. Author supplied publication, Garth Veterinary Group Duffield E Yorkshire England
Pointon AM, Surman PG, McCloud PI and White PBD (1983): 'The pattern of endemic parvovirus infection in four pig herds'. Australian Veterinary Journal 60:166-171
Sorenson KJ (1982): 'Porcine parvovirus - serological examinations in pig breeding herds'. Nordisk Veterinaermedin 8 34:329-333. Abstract in Pig New and Information (1983) 4:199
Stein TE and Leman Ad (1982): 'Epidemiologic and economic analysis of parvovirus infection in swine:. In Proceedings 7th Int. Pig Vet. Soc. Congr. Mexico City Mexico. Page 197
Too HL and Love RJ (1985): 'Persistence of passive immunity to porcine parvovirus' Australian Veterinary Journal 62 (8):282-285
Vannier P, Cariolet R and Tillion JP (1982): 'Serological survey of a parvovirus infection in a pig herd. Circumstances of the onset of reproductive disorders.' In Proceedings 6th Int. Pig Vet. Soc. Congr. Mexico City Mexico. Page 190
Vannier P, Tillon JP, Cariolet R and Madec F (1984): 'Seroepizootiological study of parvovirus in pig herds.' Zentral Vet. 31 (1):36-45. Abstract in Vet. Bulletin (1985) 56:469
Wrathall AE (1984): 'Infectious disorders of Porcine Reproduction.'
Diagnosis and Control Vet. Rec. 100:230-237.