
CASE NOTES
Aspects of yersiniosis; what happened in 2022 in NSW
Erika Bunker, Elizabeth Macarthur Agricultural Institute (EMAI), Menangle NSW
Posted Flock and Herd July 2023
Introduction
In the unusual winter season of 2022, characterised by persistent wet weather and flooding in many parts of NSW, a peak in outbreaks of yersiniosis was experienced by field veterinarians and EMAI pathology and bacteriology teams.

Image 1. Cattle surrounded by flooded paddocks, Menangle NSW, July 2022
Case distribution of yersiniosis 2006 - 2022

Image 2. Yersinia case distribution from March 2006 to December 2022. Graphs: Anthony Chamings
The graphs (Image 2) show numbers of submissions to the EMAI veterinary laboratory per month with the diagnostic codes of 'Yersinia pseudotuberculosis' and 'Yersinia enterocolitica' in our laboratory information management system over the specified years, for all animal species, but mainly cattle and sheep. The graphs reflect the seasonality of the disease and the prominent spike in case numbers in winter 2022. On a single day in August, submissions from six properties from the Hunter region, Mid and North Coast and Northern Tablelands were received with a history of several cattle found dead or sick with diarrhoea, in muddy areas, flooded paddocks or river flats previously inundated by water; one private practitioner described a cow with profuse diarrhoea he sampled as "the same as all the other cows" he had been seeing over the past month on the coast, reflecting the extent of the outbreak. In all six submissions, yersiniosis was confirmed by culture of Y. pseudotuberculosis in a profuse growth from faeces or fresh intestine.
Aetiological agent - Yersinia
Yersinia is named after the French bacteriologist, Alexandre Yersin, who in 1894 discovered Pasteurella pestis, now Yersinia pestis, the causative agent of plague. It was independently isolated in the same year by Japanese physician and bacteriologist Kitasato Shibasaburo (Britannica website).
Y. pestisis categorised into three subtypes or biovars; Antiqua, Medievalis, and Orientalis, each associated with a major pandemic. Biovar Orientalis is responsible for the current pandemic, the modern plague.
It is believed thatY. pestis evolved fromY. pseudotuberculosis a few thousand years ago. This means that it has evolved rapidly from being a pathogen widely found in the environment, to a blood-borne pathogen of mammals that can parasitise insects, with limited capacity for survival outside a host (Microbewiki website).
Since the 1990s, most human cases of modern plague have occurred in Africa. The three most endemic countries are the Democratic Republic of Congo, Madagascar and Peru. In Madagascar cases of bubonic plague are reported nearly every year during the epidemic season (WHO website).

Image 3. Plague cases worldwide 2000-2018 (Barbieri et al 2021)
Zoonotic potential of Yersinia spp.
Plague is a zoonosis, and so is non-plague yersiniosis.
Plague can be transmitted from sheep to humans - this has happened in Tibet, not in Australia. The Tibetan plateau is a natural plague endemic area. Marmots (Marmota himalayana) are the primary host and transfer the disease to sheep, probably by contact with the dead rodents or via fleas. Tibetan sheep have a habit of licking dead rodents such as marmots. Multiple human plague outbreaks have occurred, resulting in many deaths. All index cases had a history of slaughtering or skinning diseased or dead Tibetan sheep (Dai et al. 2018).
Fortunately, we only have non-plague Yersinia in Australia. Our pathogenic species are limited to Y. pseudotuberculosis and Y. enterocolitica.
Other species include Y. intermedia, Y. frederiksenii, Y. kristensenii, and several other atypical Yersinia, whose pathogenicity in humans and animals is not clearly established but may cause opportunistic infections (Smegmo et al. 1999). And then, for completeness, there is Y. ruckeri, which has been diagnosed in the past at EMAI as an aetiological agent in diseased fish.

Image 4. Diseases caused by Yersinia spp. (Markey et al. 2013)
The table (Image 4) illustrates the large spectrum of diseases and species associated with Yersinia spp.
Looking at zoonotic potential of Y. pseudotuberculosis and Y. enterocolitica is somewhat complicated due to the many bio- and serotypes and their variations in pathogenic potential for the different species of animals and humans (McNally et al. 2004).
Enteric yersiniosis in ruminants in Australia is most commonly caused by Y. pseudotuberculosis type III (which has also been isolated from humans), and Y. enterocolitica biotype 5, serotype 2 and 3 (not common in humans) (Slee et al. 1988, Slee et al. 1990, Callinan et al. 1988, Philbey et al. 1991, Slee et al. 1992, Stanger et al. 2018). Y. enterocolitica strains from ruminants are generally non-pathogenic for humans. Pigs are the major reservoir for human pathogenic strains of Yersinia spp. (Smego et al. 1999, McNally et al. 2004). Y. pseudotuberculosis type III, the most common strain in ruminants, is also common in pigs, so ruminants as well as pigs could potentially be a source of infection in humans (Slee et al. 1988).
Disease investigation of suspect yersiniosis cases
1. History

Image 5. Cattle in muddy and flooded environment, Menangle, NSW, July 2022
As mentioned in the introduction, in various 'Flock and Herd' papers by Local Land Services (LLS) veterinarians (Poe 2012, Kemsley 2012, Gillan 2016, Ellem 2022, Thomson 2011, Ball 2010) and by others (Callinan 1988, Philbey 1991), yersiniosis typically affects ruminants in the winter and spring in muddy and flooded environments. Although cases are often seen in low lying country in wet wintery conditions, "top of the mountain, million-dollar view" paddocks can also be affected if wet enough (Kemsley 2012). Dry feed exposed to mud and silt covering paddocks adjacent to previously flooded rivers are observed as a potential source of contamination.
2. Clinical signs
Scours in cattle, sheep and goats is the most common disease presentation, however in 2022, "found dead" was also a relatively common scenario. In addition to enteric disease in cattle and small ruminants, we had other disease syndromes present and other animal species affected. For example, abortions in sheep, disseminated organ lesions in alpacas (consistent with findings of Ragno et al. 2019), and a case of yersiniosis in birds. An interesting case of multiple ovine stillbirths on the Northern Tablelands, investigated in the wet 2022 season by LLS and the EMAI veterinary laboratory, had a preceding history of enteric yersiniosis on the farm, and both Y. pseudotuberculosis and Y. enterocolitica were involved (Parsons 2023).
3. Gross Pathology
In the winter of 2022, marked gross lesions of thickened and reddened guts with fibrino-necrotic exudate were often reported, however, extent of gross lesions can vary. The photos (Images 6-8) show lesions from NSW cases from earlier years (Ellem 2022, Kemsley 2012, Ball 2010).

Image 6. Colitis in a goat with Yersiniosis (Photo: Judy Ellem)

Image 7. Caecum in a cow with Yersiniosis (Photo: Phil Kemsley)

Image 8. Intestines from cow with Yersiniosis (Photo: Matt Ball)
4. Histopathology
Histology is very useful to diagnose yersiniosis, as the microabscesses found in the intestinal mucosa are almost pathognomonic. They consist of colonies of small coccobacilli surrounded by neutrophils. The lesions can be segmental, so it is worthwhile to submit several gut samples from various sites.

Image 9. Small intestine, cow with Y. pseudotuberculosis infection, H&E, 20x
Image 9 shows a section of small intestine from a cow with diarrhoea, one of several, that died in a paddock with a 'boggy' creek on the north coast of NSW in June 2022. Y. pseudotuberculosis was isolated.

Image 10. Liver, alpaca with Y. enterocolica infection, H&E, 20x
Image 10 is of a liver from an alpaca, one of several experiencing sudden death in the Central Tablelands in September 2022. Y. enterocolitica was isolated in a profuse growth from the liver.
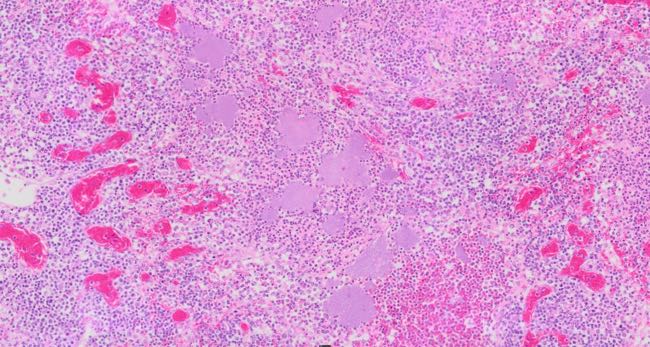
Image 11. Lymph node, alpaca with Y. enterocolica infection, H&E, 10x
Image 11 shows the lymph node from the same alpaca as Image 10, with extensive bacterial colonies.
If we only have samples from a live animal, or if we wish to confirm a histological diagnosis of Yersiniosis, we set up bacterial cultures.
5. Bacteriology
Suitable samples include faeces, fresh intestine or intestinal contents (for enteric disease) and occasionally others (for example abortion samples and organ lesions).
Yersinia are gram negative, facultative anaerobic coccobacilli that can multiply in water at a low temperature.

Image 12. Culture plates of Y. pseudotuberculosis and Y. enterocolitica (Photo: EMAI Bacteriology Team)
The culture process at EMAI involves incubation on selective Yersinia agar plates at 30 degrees. Colonies are pink (Image 12). Y. enterocolitica grows faster than Y. pseudotuberculosis and colonies look a little different - they are larger with a transparent border. Suspect colonies are subcultured. A MALDI Biotyper can then be used on pure subcultures to identify Y. pseudotuberculosis and Y. enterocolitica.
Molecular tests are available but not routinely used at EMAI for diagnostic cases; they are more useful for research purposes for typing and determining virulence genes.
Our culture results reflect that Y. pseudotuberculosis is the species that causes disease in cattle (with rare exceptions), while sheep and goat yersiniosis can be caused by both Y. pseudotuberculosis and Y. enterocolitica. Y. pseudotuberculosis is considered more pathogenic than Y. enterocolitica, commonly producing typical histological lesions, while Y. enterocolitica often only produces non-specific changes (Callinan et al. 1991).
Our results also reflect the seasonality of disease with isolation of Y. pseudotuberculosis in winter to early spring, and Y. enterocolitica all year round (Slee et al. 1992, Stanger et al. 2018). Rarely we isolate atypical Yersinia.
Diagnostic dilemma
Looking at the relatively simple diagnostic steps just described, it appears that yersiniosis is a straightforward diagnosis - you have suitable environmental conditions, scouring animals, gross gut lesions, typical microabscesses on histology, profuse growth of Yersinia species on bacterial culture, and you have your diagnosis - yersiniosis. However, things are often more complicated.
In many instances, firstly, it needs to be decided if the diagnostic criteria are sufficient to justify a diagnosis of yersiniosis, secondly if yersiniosis is the primary condition. As Yersinia is commonly isolated from healthy animals (Hodges et al. 1985, Yang et al. 2016), other supportive evidence is needed in addition to culture results.
History, background of other cases in the area and typical gross enteric lesions with a positive culture result were often enough to be confident with the diagnosis in the 2022 outbreaks.
In other instances, histology can be valuable for a definite diagnosis, although typical microabscesses are not always found. If this is due to lesions being segmental or completely absent, is sometimes unclear. It is worthwhile submitting a range of intestinal samples particularly those with gross lesions. In a study by the old Wagga Wagga Veterinary Laboratory (Philbey 1991), in the absence of other lesions or causative agents, yersiniosis was still considered the cause of diarrhoea in sheep which had no typical microabscesses, but a profuse growth of Yersinia on culture. Vise versa, in studies from Victoria, experimental infection of calves and lambs produced colonisation and typical lesions but no clinical disease (Slee et al. 1988, et al. 1990).
Even if there is sufficient evidence that you have a case of clinical yersiniosis - what else could be contributing to disease? In the literature, the role of parasites and trace elements in association with Yersinia are discussed (Ball 2010, McLennan et al. 2000, et al. 1990, Philbey et al. 1991).
Parasitism may be the primary condition or a predisposing factor. We had several cases at EMAI Veterinary Laboratory where, apart from a positive Yersinia culture, there was also evidence of a worm burden, coccidia, liver fluke or stomach fluke, or a combination of these. Mineral deficiencies also need to be considered. High rainfall and lush pastures favour selenium deficiency. We have tested for selenium (via glutathione peroxidase) and copper in multiple bovine Yersinia submissions and found low selenium levels, low copper levels or both. Poor nutrition is another factor. As Ball (2010) noted, winter in the north coast areas coincides with a decline in pasture availability. Other stress factors include inclement weather, transport, pregnancy, lactation and weaning. Of cases submitted to EMAI over the years, some scouring bovines with Y. pseudotuberculosis cultured also had Salmonella isolated. One was also a persistent Pestivirus carrier, and another had a positive Johne's culture.
Apart from Yersinia, weighing up multiple factors, particularly mineral and parasite status, is important for management. In the end, Yersinia is there, and you can't eliminate it easily. It is shed by carriers and survives for long periods in the environment. One can attempt to reduce the level of exposure to Yersinia by identifying and eliminating or controlling other predisposing or concurrent factors, especially the most common ones like mineral deficiency and parasitism.
As an example, histological sections are presented below from two cases requiring careful assessment of the parasite status versus Yersinia significance to decide on the primary diagnosis.

Image 13. Small intestine, cow, with a coccidial schizont adjacent to colonies of Y. pseudotuberculosis forming a microabscess, H&E, 20x
The first section (Image 13) is small intestine from a cow with diarrhoea, with typical microabscesses and a coccidial schizont placed next to a microabscess. Y. pseudotuberculosis was isolated in a profuse growth. Not many coccidia were present, and yersiniosis was considered the primary condition.

Image 14. Small intestine, sheep, H&E, 10x
The other section (Image 14) is small intestine from a sheep with diarrhoea, and Y. enterocolitica was isolated in a profuse growth. No microabscesses were seen, but many nematodes in several histological sections. Faecal egg count (FEC) was high. Parasitism was considered the primary condition.
Another interesting and more complex 2022 case study included a property in the lower Hunter Valley where many cattle were affected. In August 2022, 60 of 120 one-year-old beef cattle were sick and ill-thrifty, some with diarrhoea. Yersinia was isolated from most of 10 faecal samples. Serum copper levels were low in the majority of 30 blood samples. Pooled FECs ranged between 320 and 1240 eggs per gram; larval culture revealed a mixed burden including Cooperia, Haemonchus, Ostertagia, and Trichostrongylus species. Liver fluke eggs and high numbers of stomach fluke eggs (several hundred) were also detected in most pools. The low copper levels, worm burden, liver fluke and stomach fluke burdens and Yersinia infection could all have contributed to ill-thrift and diarrhoea in these cattle.
In late September 2022, 11 faeces from cattle on the same property were submitted for monitoring of Yersinia. No clinical disease was noted in these cattle. We isolated Y. pseudotuberculosis from most of them.
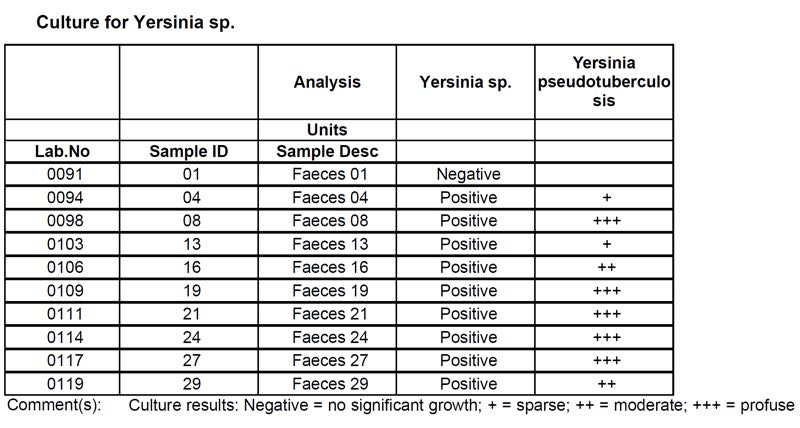
Image 15. Yersinia culture results of diseased cattle from the property in the lower Hunter Valley, August 2022

Image 16: Yersinia culture results from clinically normal cattle on the same property in the lower Hunter Valley, September 2022
The tables above (Image 15, 16) show the August and September culture results. There appears to be a slight reduction in numbers of colonies grown in the healthy cattle sampled in September compared to the diseased cattle sampled in August, however, Y. pseudotuberculosis is clearly still present in the absence of disease. These results are not surprising, as Y. pseudotuberculosis can be isolated from healthy cattle. A New Zealand study found that Y. pseudotuberculosis type III infection was common in 8- to 13-month-old cattle (Hodges et al. 1985). Excretion also tends to persist for a while. In sheep, Y. pseudotuberculosis persisted for up to 14 weeks after experimental infection; Y. enterocolitica even longer for up to 29 weeks (Slee et al. 1992).
Conclusion
It is advised to watch local environmental conditions and be prepared. Ideal conditions are expected sometime after heavy rainfall and flooding, to give Yersinia time to multiply in anaerobic moist conditions, for example in 2009 outbreaks in cattle at the NSW north coast occurred in July and August after flooding events in late May (Ball 2010). This means there is often time to implement some preventative measures to avoid yersiniosis. One could check the parasite and mineral status of animals before they get sick. Unfortunately, parasites and selenium deficiency occur in the same conditions, namely in wet and lush seasons.
If animals get sick in the wet, gather and evaluate enough diagnostic criteria, not only limited to clinical evidence of diarrhoea and culture results. It is recommended to submit a good range of samples to not only test for yersiniosis via bacteriology and histopathology, but also assess for other diseases that can cause diarrhoea or predispose to yersiniosis.
References
- Ball M (2010) Pathology and Epidemiology of Bovine Yersiniosis. Posted on Flock & Herd August 2010 www.flockandherd.net.au
- Barbieri R, Signoli M, Chevé D, Costedoat C, Tzortzis S, Aboudharam G, Raoult D & Drancourt M (2021) Yersinia pestis: the natural history of plague Clinical Microbiology Reviews 34:e00044-19
- Britannica website www.britannica.com
- Callinan RB, Cook RW, Boulton JG, Fraser GC & Unger DB (1988) Enterocolitis in cattle associated with Yersinia pseudotuberculosis infection Australian Veterinary Journal 65:8-11
- Dai R, Wei B, Xiong H, Yang X, Peng Y, He J et al. (2018) Human plague associated with Tibetan sheep originates in marmots PLoS Neglected Tropical Diseases 12(8):e0006635
- Ellem J (2022) Yersinia enterocolitica enteritis in a goat herd. Posted on Flock & Herd May 2022 www.flockandherd.net.au
- Gillan N (2016) Yersinia pseudotuberculosis enteritis in adult sheep. Posted on Flock & Herd March 2016 www.flockandherd.net.au
- Hodges RT & Carman MG (1985) Recovery of Yersinia pseudotuberculosis from the faeces of healthy cattle New Zealand Veterinary Journal 33(10)175-176
- Kemsley P (2012) Yersiniosis in cattle. Posted on Flock & Herd September 2012 www.flockandherd.net.au
- Markey BK et al. (2013) Diseases caused by Yersinia species Clinical Veterinary Microbiology (Second Ed), Mosby Elsevier, pp 267-269
- McLennan MW & Kerr DR (2000) Yersiniosis and trace element deficiency in a dairy herd Australian Veterinary Journal 78(1):28-30
- McNally A, Cheasty T, Fearnley C, Dalziel RW, Paiba GA, Manning G & Newell DG (2004) Comparison of the biotypes of Yersinia enterocolitica isolated from pigs, cattle and sheep at slaughter and from humans with yersiniosis in Great Britain during 1999-2000 Letters in Applied Microbiology 39:103-108
- Microbewiki website microbewiki.kenyon.edu
- Parsons M (2023) Yersiniosis causing stillbirths in maiden ewes. Posted on Flock &Herd March 2023 www.flockandherd.net.au
- Philbey AW, Glastonbury JRW, Links IJ & Matthews LM (1991) Yersinia species isolated from sheep with enterocolitis Australian Veterinary Journal 68:108-110
- Poe I (2012) Yersinia enterocolitica abortion in a doe. Posted Flock & Herd September 2012 www.flockandherd.net.au
- Ragno VM, Uehlinger FD, Gabadage K, Movasseghi AR & Montgomery JB (2019) Investigation of a Yersinia enterocolitica outbreak in a commercial alpaca farm in Saskatchewan Canadian Veterinary Journal 60:877-882
- Slee KJ, Brightling P & Seiler RJ (1988) Enteritis in cattle due to Yersinia pseudotuberculosisminfection Australian Veterinarian Journal 65:9
- Slee K & Button C (1990) Enteritis in sheep, goats and pigs due to Yersinia pseudotuberculosis infection Australian Veterinary Journal 67:320-322
- Slee KJ & Button C (1990) Enteritis in sheep and goats due to Yersinia enterocolitica infection Australian Veterinary Journal 67:396-398
- Slee KJ & Skilbeck NW (1992) Epidemiology of Yersinia pseudotuberculosis and Y. enterocolitica Infections in Sheep in Australia Journal of Clinical Microbiology 30(3):712-715
- Smego RA, Frean J & Koornhof HJ (1999) Review. Yersiniosis I: Microbiological and Clinicoepidemiological Aspects of Plague and Non-Plague YersiniamInfections European Journal of Clinical Microbiology and Infectious Diseases 18:1-15
- Stanger KJ, McGregor H & Larsen JWA (2018) Outbreaks of diarrhoea ('winter scours') in weaned Merino in south-eastern Australia Australian Veterinary Journal 96:176-183
- Thomson D (2011) Yersiniosis or more? Posted on Flock & Herd October 2011 www.flockandherd.net.au
- WHO website www.who.int
- Yang R, Ryan U, Gardner G, Carmichael I, Campbell AJD & Jacobson C (2016) Prevalence, faecal shedding and genetic characterisation of Yersinia spp. in sheep across four states of Australia Australian Veterinary Journal 94:129-137