
CASE NOTES
Hepatic Coccidiosis in Meat Rabbits
Tania Areori, Charles Sturt University Final Year Veterinary Science Student, and Liz Bolin, District Veterinarian, North Coast Local Land Services
Posted Flock & Herd November 2020
Introduction
This report describes an outbreak of hepatic coccidiosis in 2019 that led to the death of 19 out of 21 mixed-breed meat-producing rabbits in Dorroughby, northern NSW. The case provides a good example of the importance of agent, host and environment factors that collectively play an important role in the occurrence of the disease.
History
The farmer runs a five-hectare property in Dorroughby, northern NSW. He was rearing pigs and ventured into meat-producing rabbits, starting with 21 New Zealand White, Giant Flemish and mixed-breed juvenile rabbits. They were kept in a cage and fed a regular rabbit-formulated diet and had access to clean fresh water. Within five weeks of arrival the owner reported that the rabbits' behaviour had changed. They were observed to be lying down on their abdomens and, after only a short illness, would die. Over a period of two weeks he lost seven out of the original 21 rabbits.
Post-mortem findings
A dead rabbit was presented to the District Veterinarian (DV), Lismore. On post-mortem, the significant findings included clear yellow peritoneal fluid, discolouration and congestion of the lungs, and multi-focal white necrotic lesions on the liver. Other findings included cloudy urine but normal small and large intestines. Based on history and post-mortem findings, differential diagnoses include rabbit hemorrhagic disease virus (RHDV) type 1 and 2, and tularaemia. Samples taken included liver, pancreas, spleen, lung, kidney, brainstem and cerebellum tissues, both fresh and fixed in formalin for histopathology and RHDV testing. Fresh liver samples were sent for RHDV antigen ELISA and PCR.
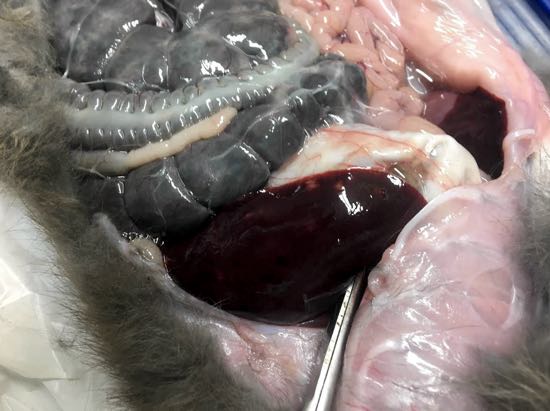
Image 1. Liver showing multifocal white necrotic lesions

Image 2. Clear yellow peritoneal fluid in the abdomen on post-mortem
Laboratory findings
The PCR and ELISA results were negative for RDHV type 1 and 2. Histopathologic examination of the liver indicated that the bile ducts were severely ectatic, with the bile duct epithelium hyperplastic, forming papillae that extended into the lumen. The epithelium contained numerous coccidial forms in various stages of development, and the lumen contained oocysts and cellular debris. The periductal stroma was infiltrated by lymphoplasmocytic cells and macrophages, with excessive fibrosing tissue and oedema in some areas. There was compression of hepatic parenchyma adjacent to these lesions. Less affected portal areas had mild infiltrates of lymphoid cells.
The spleen showed a white pulp prominent with areas of apoptosis or necrosis. In the white pulp there were significant macrophages containing droplets or clumps of yellow-brown pigment. The lung showed marked congestion and, similarly, the kidney was moderately congested.
There were no significant findings in the heart, pancreas, and brain samples. Based on the histological findings, hepatic coccidiosis was diagnosed.
Treatment
Advice from the Brisbane Bird and Exotics Veterinary Service (BBEVS) indicated that treatment of hepatic coccidiosis in rabbits is off label. Treatment options include trimethoprim sulpha 30mg/kg PO daily for 10 days; sulfadimethoxine 50mg/kgPO first dose, then 25 mg/kg every 24hr for 10-20 days; or amprolium 9.6% in drinking water (0.5ml per 500ml). According to advice from the vet at BBEVS, amprolium is not consistently effective as water consumption can be variable.
There is no WHP information for meat rabbits.
Unfortunately, despite treatment, only two rabbits survived.
Discussion
Rabbit coccidiosis or Eimeriosis is caused by the apicomplexan protozoan parasiteEimeria spp.and is an important primary causative agent in digestive disease in meat rabbits (Hamid, Prastowo, & Kristianingrum, 2019). These protozoan parasites are ubiquitous, found naturally in the environment. They are spread via the faecal-oral route between infected and naïve populations. There are two forms of coccidiosis recognised in rabbits, hepatic and intestinal (Sivajothi, Reddy, & Rayulu, 2016).Eimera sitadaeis the primary coccidian causing hepatic coccidiosis in rabbits.
A review of the literature shows that regardless of rabbit species or whether wild and domestic, there is a large prevalence of natural infection occurring in rabbit populations by multipleEimeriaspecies (Duszynski & Couch, 2013). Rabbit coccidiosis is a common cause of disease in juvenile rabbits. Adults can be infected but usually do not show clinical signs of infection - instead they act as carriers and discharge oocysts into the environment (Duszynski & Couch, 2013). Severity of infection is dependent in large part on the number of oocysts ingested, the coccidian species and its specific pathophysiology, the host immune system, and rabbit husbandry (Duszynski & Couch, 2013). Animal husbandry, group housing and stress are key risk factors for clinical disease, especially for young vulnerable rabbits that are just weaned as identified by Okumuet al.(2014). A study by Papeschi, Fichi, & Perrucci, 2013, found that juvenile rabbits had a high number of oocysts and attributed this finding to the incomplete maturity of their immune system and inadequate immune responses mounted to previous infections. The rabbits affected in this case study were all weaned juveniles.
Rabbits infected with hepatic coccidiosis have varying presentations depending on the severity of infection. Mild infections show no clinical signs whereas in moderate infections, poor growth rate is observed. In severe infections, there is loss of appetite, weakness, and constipation in the later stages of the disease (Sivajothiet al.2016). There was also a study where rabbits presented with anorexia, debilitation, diarrhoea, icterus, rough hair coat and pendulous abdomens (Al-Taee, Al-Naimi, Khalaf, & Tano, 2012).
The clinical signs of hepatic coccidiosis reflect the pathophysiology of the disease. The life cycle ofEimeriatakes 4-14 days with the start of the cycle initiated by ingestion of infected faecal material containing oocysts. The digestive enzymes in the intestines result in rupture of the oocysts which release sporozoites, which then attach to intestinal, hepatic and bile duct epithelium, where the oocysts undergo merogony (i.e. asexual division) to produce merozoites, which then mature and rupture from the host cell. These merozoites then infect new cells to repeat merogony, which then leads to the sexual reproductive stage. The infection and subsequent rupture of the host cells leads to cell damage and depending on the tissue affected, clinical signs will manifest accordingly (Hall, 2012).
Diagnosis of coccidiosis in live rabbits is difficult. Faecal examination for oocysts is one method, however it is difficult to identify theEimeriaspecies based on faecal examination and to differentiate the from the rabbit-specific yeast,Cyniclomyces guttulatus (Hall, 2012).
The post-mortem in this case showed focal necrotic lesions on the liver and a clear yellow peritoneal fluid. Sivajothi (2016) found similar post-mortem findings on rabbits suspected of having hepatic coccidiosis, which revealed 1-5mm white nodules spread over the hepatic surface and distension of the bile ducts. Al-mathal (2008) also observed similar changes to the liver on post-mortem and observed serous exudate in the peritoneal cavity.
Histopathological changes reported have included severe congestion and dilation of central veins, rupturing of the endothelial lining, hyperplasia of the epithelial lining of portal areas with finger-like projections in the lumen of the bile duct, congestion and dilation of sinusoids with haemorrhagic areas. Multiple areas of coagulative necrosis of hepatic cells surrounded with inflammatory cells were also found (Al-mathal, 2008). Similarly, Kim, Reilly, Schommer, and Spagnoli (2010) reported biliary hyperplasia, interepithelialEimeriacysts and necrotising multifocal hepatitis. The histological changes reported in the literature are consistent with those found in this case.
Çam et al. 2008 reports that treatment with toltrazuril and a toltrazuril/ivermectin combination were highly effective in reducing faecal oocyst output in infected rabbits. Redrobeet al. 2010 did a comparison of both sulphonamide and toltrazuril on infected rabbits and both were highly effective with 73-99% elimination of the protozoa. However, species other than those in the genusEimeriawere also included in this study.
Prevention of coccidiosis has been described by several studies and includes good animal husbandry, anti-coccidial treatment with feed and water, avoidance of early weaning, quarantining introduced animals and avoiding stress during high-risk periods (Okumuet al.2014; Papeschiet al.2013).
Conclusion
Hepatic coccidiosis is a common infection, especially in juvenile rabbits. This case study shows how the interaction between environment, host and pathogen factors results in the expression of disease. Farmers and veterinarians are able to influence the role of the environment through changes to husbandry.
Acknowledgements
Dr Luzia Rast for organising practice rotation.
References
- Al-mathal EM (2008). Hepatic coccidiosis of the domestic rabbit (Oryctolagus cuniculus domesticus) in Saudi Arabia. World Journal of Zoology 3(1);30-35
- Al-Taee EH, Al-Naimi RAS, Khalaf OH & Tano SY (2012). Pathological study of hepatic coccidiosis in naturally infected rabbits. Al-Qadisiyah Journal of Veterinary Medicine Sciences 11(1);63 doi.org
- Çam Y, Atasever A, Eraslan G, Kibar M, Atalay Ö, Beyaz L, & Liman BC (2008). Eimeria stiedae: Experimental infection in rabbits and the effect of treatment with toltrazuril and ivermectin. Experimental Parasitology 119(1);164-172 doi: doi.org
- Duszynski DW & Couch L (2013). The biology and identification of the Coccidia (Apicomplexa) of rabbits of the world. Saint Louis, United States: Elsevier
- Hamid PH, Prastowo S & Kristianingrum YP (2019). Intestinal and hepatic coccidiosis among rabbits in Yogyakarta, Indonesia. Veterinary world 12(8);1256-1260 doi.org
- Kim DY, Reilly TJ, Schommer SK & Spagnoli ST (2010). Rabbit tularemia and hepatic coccidiosis in wild rabbit. Emerging infectious diseases 16(12);2016-2017 doi.org
- Okumu PO, Gathumbi PK, Karanja DN, Mande JD, Wanyoike MM, Gachuiri CK, & Borter, D. K. (2014). Prevalence, pathology and risk factors for coccidiosis in domestic rabbits (Oryctolagus cuniculus) in selected regions in Kenya. Veterinary Quarterly 34(4);205-210 doi.org
- Papeschi C, Fichi G, & Perrucci S (2013). Oocyst excretion pattern of three intestinal Eimeria species in female rabbits. World Rabbit Science 21(2);77-83 doi.org
- Redrobe SP, Gakos G, Elliot SC, Saunders R, Martin S & Morgan ER (2010). Comparison of toltrazuril and sulphadimethoxine in the treatment of intestinal coccidiosis in pet rabbits. The Veterinary Record 167(8);287 doi.org
- Sivajothi S, Reddy B, & Rayulu V (2016). Study on impression smears of hepatic coccidiosis in rabbits. Journal of Parasitic Diseases 40(3);906-909 doi.org
- van Praag, E. Protozoal enteritis: Coccidiosis. www.medirabbit.com Retrieved 1 November 2020