
CASE NOTES
Q FEVER: A NOTIFIABLE DISEASE IN HUMANS
Ian Lugton, SDV, SELHPA, Bega and Anthony Brown, A/Prof, Sydney University Medical School, Dubbo
Posted Flock & Herd March 2011
History
E. H. Derrick first reported in 1937 on ?Query Fever?. The disease first identified in Brisbane abattoir workers is now known as Q fever. Derrick sent the organisms isolated to McFarlane Burnet. Burnet and Freeman recognised organism as a Rickettsia. Davis and Cox studying ?Nine Mile disease? in Montana, reported the isolation of this organism from ticks in 1938. The organism was called Coxiella burnetii in 1948, in recognition of these pioneering researchers.
THE ORGANISM
C. burnetii is a member of the Rickettsiaceae. It is an obligatory intracellular gram-negative, rod-shaped bacterium. The organism is moderately resistant to heat, but not to pasteurisation. It survives well in air, soil and water. In dust it will survive for over a year and can be readily disseminated on fomites such as wool, hides, clothing, straw and packing materials.
The organism is taxonomically grouped with Legionella and Francisella. Its life cycle is completed within acidic phagolysosomes of macrphages/monocytes.
INFECTIVITY
In the host animal, C. burnetii organisms are most abundant in foetal/placental fluids and tissues at concentrations up to 109/gram of tissue. These concentrations are more than one billion times the Infectious Dose 50.
Only a few organisms are required to establish infection – mostly by the respiratory route. It has been speculated that as few as one organism inhaled into the lungs is required to cause infection.
Most infections are acquired by contacting infected tissues, or contaminated items or aerosols and dusts from these. Ticks are believed unimportant in transmission.
C. burnetii organisms have two life cycle stages: The large-cell variant (LCV) vegetative form seen in infected monocytes and macrophages and the small cell spore-like variant (SCV). The SCV is found extracellularly and is the infectious form of the organism. The ?spore? form survives exposure to physical and chemical disruption.
THE DISEASE
Q Fever in humans usually presents as an acute illness characterised by rapid onset of high fever, rigors, profuse sweats, extreme fatigue, muscle and joint pain, severe headache and photophobia. The incubation period is normally 14 to 21 days, but can be as short as 4 days and as long as 6 weeks.
Often sufferers will show some evidence of hepatitis and in more severe cases can become jaundiced. Mild pneumonia is another common sequel to infection. If untreated, Q Fever symptoms last for around 1 to 3 weeks. Common differential diagnoses include influenza, leptospirosis and brucellosis.
In infected animals there are usually no clinical signs. However, late abortions and stillbirths are possible consequences of infection. The disease is occasionally diagnosed in goats and sheep. Recently in the Netherlands infected dairy goat farms have had abortion rates up to 60%.
In humans 60% of infections are asymptomatic. However, 38% experience an acute self-limiting illness from 2 to 6 weeks. Around one in 50 require hospitalisation because of complications associated with hepatitis, pneumonia or meningoencephalitis. Acute disease during pregnancy results in abortions in around 0.5% of women infected. Infection in early pregnancy, or even before conception, can recrudesce at term and cause foetal damage. A few people may die, particularly the elderly and the debilitated, with the mortality rate around 2.5%.
C. burnetii is sufficiently tractable, infectious and debilitating to have it classified as a bioterrorism agent, Category B.
Chronic disease may follow acute infections. Lesions associated with this chronicity include subacute endocarditis, which may require heart valve replacement, and granulomatous lesions in bone, joints, liver, lung, testis, or other soft tissues.
Chronic fatigue syndrome may occur in 10-15% of people following acute infections. However, the role of Q Fever in this syndrome is often difficult to determine with current testing technology.
IMMUNOLOGY AND DIAGNOSIS
To complicate diagnosis, C. burnetii has two phases of antigenic variation, related to surface lipopolysaccharide expression. Phase 1 response is associated with acute infections and antibody blocking. Such blocking may in part explain the existence of a carrier state in infected animals and humans. Phase 2 antigens are expressed in culture media. This antigenic variation is helpful in differentiating between acute and chronic Q Fever. Phase II IgM is expressed first in an infection, and phase I IgG showing later, being detected up to 8 weeks post-infection.
Cell-mediated immunity also important and this reactivity contributes to chronic granulomatous responses observed in many cases. Poor CMI responses are also likely to contribute to the development of chronic infections.
Diagnosis of infection in humans is usually based on a history which might include an episode of a flu-like illness, and on serology. There are several serological tests that may be used, including enzyme immunoassay (EIA), complement fixation (CFT) and immunofluorescence tests (IFA). False-positive (cross-reactive) IgM results are relatively common (~10-20%).
Acute and convalescent serology is recommended to aid diagnosis. The organism may be detected in blood up to day 10 after illness onset. If examined within ten days of onset of acute signs, EDTA blood can be used for PCR tests and also potentially for culture of the organism in level 3 containment facilities.
In animals there are no universally recognised or accepted serological tests. However, CFT, IFA and ELISAs have all been used.
In chronic cases, C. burnetii DNA may be detected by PCR in peripheral blood mononuclear cells or in biopsy specimens from granulomatous tissue. Immunohistochemistry has also been employed. Skin testing, through a delayed-type hypersensitivity (DTH) reaction of the CMI, can also be used to check past exposure. In some cases of chronic disease neither serology, nor skin testing will elicit positive responses.
The NSW Health Infectious Disease website has a factsheet on Q Fever that provides a stepwise approach to interpretation of serological tests.
TREATMENT OF Q FEVER
Treatment should be instigated before laboratory confirmation of Q Fever. Doxycycline 100mg b i.d for 10 days is the recommended treatment. Erythromycin, rifampicin, ciprofloxacin are also effective.
Symptomatic management of the disorder involves the use of analgesics, anti-pyretics and anti-inflammatory agents. Chronic infection may require years of antibiotic treatment with a combination of antibiotics.
EPIDEMIOLOGY
As this is a zoonosis of little or poorly recognised animal health significance, it historically has received little veterinary attention. As a consequence there is a poor understanding of the epidemiology in animals, with many published papers only reporting spectacular or unusual occurrences. However, it is a disease of interest to the medical profession and there is a fair body of information on the disease in humans. Perhaps this is an infectious organism that should be incorporated into the ?One Medicine? paradigm? This way it might receive more cross-disciplinary attention to the betterment of both the veterinary and medical professions.
C. burnetti infects wild and domestic animals and their ticks. Sheep, goats, cattle, camelids and a variety of wildlife may be infected. There is little local information about the prevalence in animals in Australia. However, the infection is also known to occur in cats, dogs and rabbits but is not recorded in pigs.
Up to 80% stock and 96% of herds/flocks can be infected in high prevalence situations. A recent review paper from the EU suggests that worldwide, there is an apparent prevalence in cattle of 20.0% and 37.7% at the animal and herd levels respectively, compared with 15% and 25% for smaller ruminants.
In animals the infection is silent until females become pregnant. This often causes a recrudescence of the infection with excretion occurring in the peri- and post-parturient periods. At this time transmission is by body fluids, faeces and milk. However, the most infectious tissues are the placenta and the birth-associated fluids.
Q Fever is mostly considered an occupational disease risk, especially in Australia, France and Spain (it is not known to occur in NZ). Some human populations are up to 40% seropositive.
Transmission to humans is mostly via infected aerosols and dust, especially under dry and windy conditions. Spread is also possible with raw milk from goats and sheep, however, transmission from such sources has not been conclusively demonstrated.
At risk workers in Australia are mostly found in the meat industry (60% of cases) and the livestock industries, including shearers and veterinarians. Others people may be exposed incidentally to stockyards, manure dust, feedlots, and processing plants by being in the vicinity or through having exposure to wind-borne dust.
In the Netherlands (and less so in Germany) there has been a major increase in human cases commencing in 2007. Since then Q fever has become a major public health problem in the Netherlands with 2,357 human cases notified in 2009 (15 cases/100,000). By comparison, in Australia in 2002 (one of our worst years) the figure was 3.9 cases/100,000.
Ongoing research in the Netherlands has confirmed that abortion waves on dairy goat farms are the primary source of infection for humans, primarily affecting people living close to dairy goat farms. Those living within 2 km of a dairy goat farm had a much higher risk for Q Fever than those living more than 5 km away (relative risk of 31.1).
To reverse the increasing incidence of Q Fever in the Netherlands, over the last three years, drastic measures have been implemented. These have included compulsory PCR testing of goat and sheep milk, compulsory vaccination of goats and sheep, and the large-scale culling of pregnant goats and infected males (62,500) on infected farms. These measures have proved successful, with only 382 human cases reported in 2010.
As a local example of the infection risk associated with large animal obstetrics, a Moruya vet practitioner in 1994 reported that 13 of 18 bystanders at a social function became infected from a contaminated calving. However, only two of those present assisted with the calving.
Small animals also present a risk. For example, a caesarean performed on bitch at a Sydney veterinary clinic several years ago resulted in several human cases. This bitch may have been a roaming scavenger and hence more likely to have had contact with C. burnetii. At the clinic, unsuccessful attempts to resuscitate the pups resulted in the practice manager becoming critically ill in hospital, a nurse sick for a week and another with malaise. One of two vet students present also became ill. However, interestingly the operating vet was unaffected, perhaps because she was wearing personal protective gear and did not handle the pups or perhaps because she had previous exposure.
There have been other similar cases in vet practices in Sydney in recent times. These incidents have caused concern and agitation for action amongst small animal practitioners, who have hitherto believed that this infection was principally associated only with large animal practice.
Cat-related reports in the international literature are common. A US epidemiological study in 1988, found that exposures to newborn animals (chiefly kittens) and stillborn kittens were significant risk factors for Q Fever. Some interesting and impressive cases have involved many human infections from a single source. Up to 33 people were infected in single village outbreak in Nova Scotia following the stillbirth of a litter of kittens and a 3 week-long bloody vaginal discharge from the cat. In another report 16/32 workers in a truck repair business, also in Nova Scotia, developed Q Fever following exposure to contaminated clothes and dust from a co-worker that had been heavily exposed at home when hand rearing kittens.
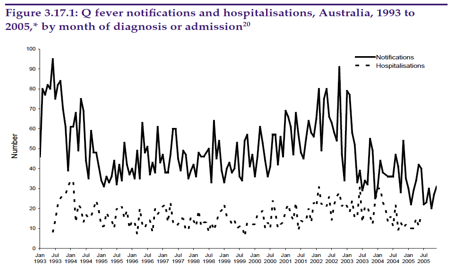
Figure 1. National Q Fever notifications from 1993 to 2005
Q Fever is arguably the most costly Australian zoonosis. For example in 2007 there were 450, 106, 92, 38, 1 and 0 cases respectively of Q Fever, Leptospirosis, Psittacosis, Brucellosis, Anthrax and ABLV notified nationally.
In Australia there have historically been around 600 Q Fever notifications annually. A National Q Fever Management Program commenced in July 2001. The program provided funding for free vaccine for people at occupational risk of Q fever. As this program has gained momentum the incidence of Q Fever has declined considerably (Figures 1 and 2).
The reported incidence is, however, likely to be an underestimate of the true situation, as the disease may be overlooked in the differential diagnostic list by many doctors. In some cases the full suite of tests available are not employed to assist in making the diagnosis. Skin testing is an obvious example.

Figure 2. NSW data following on from the national data shown in Figure 1. The pattern of steady decline in notifications from 2002 to 2005 continues up till the present time.
Q Fever occurs predominantly in working men in 20 to 50 year age group and hence has a significant impact on the paid workforce (Figure 3). WorkCover compensation has been estimated at around $1.3M annually, and some legal settlements have been for over $1M. Direct medical costs for acute disease may escalate beyond $3K.
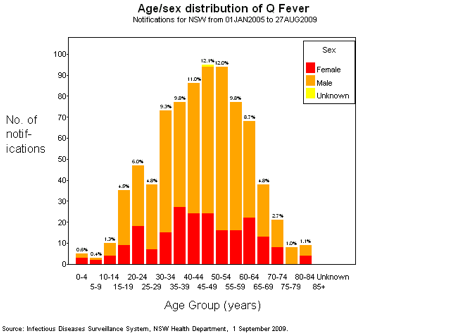
Figure 3. Chart of age by gender distribution of Q Fever in NSW between January 2005 and August 2009
Historically the disease has been most common in Queensland, followed by NSW. Around 90% of cases occur in these two states, hence the distribution shown in the map in Figure 4, which shows a higher incidence in Northern NSW. Q Fever is rare in Western Australia and the Northern Territory.

Figure 4. NSW map showing the incidence of Q Fever from 2000 to 2009
PREVENTION
Medical practitioners aiming to minimise the transmission of zoonoses advise people to avoid contact with animals. However, for many in the population this is neither desirable, nor achievable. People should, however, wash hands after contact with animals. Animal tissues must be handled hygienically, especially birth-associated tissues and fluids.
Placentae, birth products, fetal membranes, and aborted/stillborn foetuses should be disposed of appropriately to reduce risks. Stock supplying raw milk for human consumption should be considered for PCR testing in case such milk is capable of initiating human cases.
Abattoirs and saleyards should be located away from populated areas and downwind. Measures should be implemented to prevent contaminated dust blowing into populated areas.
Access to workplaces with high risk of Q fever should be restricted to workers required to be there, vaccinated people or those otherwise known to be immune.
For those in at-risk occupations protective clothing should be worn. Contaminated clothing should be laundered appropriately, taking care to avoid unnecessary family exposure. There should be no eating, drinking, smoking, or nail biting in risky situations including in animal holding facilities. The production and exposure to aerosols from animal tissues and faeces should be minimized. Respiratory protection devices (P2 masks) suitable for preventing aerosol inhalation should be used by workers at increased risk of inhaling contaminated particles.
For those at high risk, such as abattoir workers, veterinarians and farmers, vaccination is strongly advised or mandatory in some work places. Vaccination is a once only process costing around $100, with the vaccine believed to be highly effective in high risk groups.
The vaccine called Q-VAX is made by CSL Biotherapies. CSL also manufactures the antigen used in the skin test (Q-VAX Skin Test). Q-VAX is the only Q-fever vaccine available in Australia. Q-VAX is a purified suspension of formalin inactivated C. burnetii grown in the yolk sacs of embryonated eggs. It was taken off the market some years ago, however, the ensuing protest from industries at risk and lobby groups, ensured the continued production of the vaccine.
Serum antibody estimations and skin tests prior to immunisation are required for all who are to be immunised to prevent serious hypersensitivity reactions. On occasions, a history suggestive of Q Fever, even in the absence of positive tests, should be considered prior to use of the vaccine, especially if the person has a history of exposure risk.
The skin test measures a DTH reaction, similar to the Mantoux test for tuberculosis. More past exposures are detected with skin tests than with antibody tests.
Vaccination contraindications include: prior Q fever infection, even if unconfirmed in a person working in a high risk occupation; previous vaccination; positive serology or skin test reaction; allergy to eggs or other vaccine component; children under 15 years, or; in people that are immunocompromised. Vaccination is not recommended during pregnancy.
If tests show an equivocal response, retesting is recommended. Partial dose vaccinations can also be used in cases where test results are not clear and there is a history suggestive of infection.
Adverse reactions to the vaccine may include local reactions, with half of those vaccinated becoming sore at the site of injection. Around 10% will develop mild ?flu-like? symptoms. A small proportion of people may develop the Q Fever fatigue syndrome or succumb to a delayed local reaction.
The Australian Q Fever Register, in place since 2002, is a database to store information about the Q Fever immune status of people, who have been tested and who have agreed to be part of the register. The purpose of the Register is to help people working in high risk industries avoid the disease. It does this by allowing employers to quickly determine an individual?s immune status and therefore need for vaccination prior to exposure. The register also holds records on vaccinations, diagnoses, and screening test results.