
CASE NOTES
Obstructive rhinopathy associated with the consumption of grass contaminated with the sporidesmin toxin from the fungus Pseudopithomyces chartarum
Héctor Ruiz1, Pablo Quílez1, Marta Ruiz de Arcaute1, Marcelo de las Heras1, Ane Garziandia2, Ane Rivas3, Enrique Castells4, David Guallar1, Delia Lacasta1
- Ruminant clinical service of the Department of Animal Pathology and Instituto Agroalimentario de Aragón-IA2
(Universidad de Zaragoza-CITA), Veterinary Faculty, University of Zaragoza, Zaragoza, 50013, Spain
- Gomiztegi koop. Basque Country. Spain
- Veterinary Practitioner. Basque Country. Spain
- Centro clínico Veterinario. Zaragoza. Spain
Posted Flock and Herd March 2026
SUMMARY
In 2022, six Blonde-faced Latxa sheep from the Basque Country (Spain) were referred to the Ruminant Clinical Service (SCRUM) for severe respiratory distress and persistent snoring, affecting up to 70-90% of animals in some flocks. Clinical examination revealed bilateral serous nasal discharge and inspiratory dyspnoea. Biochemistry showed hepatic and pancreatic alterations, and CT imaging identified soft-tissue obstruction of the rostral nasal cavity at the alar fold level. Necropsy confirmed chronic hepatic atrophy and fibrosis consistent with pseudopithomycotoxicosis, together with whitish proliferative lesions of the alar folds. Histopathology revealed moderate-to-severe arteriosclerosis of small nasal arteries, similar to vascular lesions observed in the liver. Pseudopithomyces chartarum was detected in pasture samples. These findings support a direct effect of sporidesmin on the rostral nasal cavity as an atypical presentation of pseudopithomycotoxicosis in sheep.
INTRODUCTION
Facial eczema (FE), also called pseudopithomycotoxicosis, is a well-recognised mycotoxicosis of grazing ruminants caused by sporidesmin, a hepatotoxic mycotoxin produced by Pseudopithomyces chartarum under warm and humid environmental conditions. The disease has been widely described in sheep, particularly in regions with high rainfall, as in New Zealand, and is classically characterised by hepatic damage, secondary photosensitisation and cutaneous lesions affecting poorly pigmented areas (Dingley, 1962; Di Menna et al., 2009). Although the hepatic and dermatological manifestations of FE are well documented, the spectrum of clinical presentations associated with chronic exposure to sporidesmin may be broader than currently recognised.
During 2021 and 2022, veterinary practitioners in the Basque Country (northern Spain) reported an unusual clinical problem affecting several sheep flocks, characterised by a high prevalence of respiratory distress, affecting more than 70% of animals in some flocks. The most striking clinical sign was continuous and intense snoring, suggestive of upper respiratory tract involvement. In a smaller proportion of animals, facial skin lesions were also observed. Despite the high morbidity, milk production was not reported to be significantly affected and farmers primarily expressed concern about the respiratory signs.
Given the consistency of clinical findings across flocks and the lack of an evident aetiology, six sheep from different affected flocks were referred to SCRUM for further investigation. All animals belonged to the Blonde-faced Latxa breed, although similar clinical signs had also been observed in Black-faced Latxa flocks. Latxa sheep are an autochthonous indigenous dairy breed reared under semi-intensive grazing systems in a region characterised by high humidity, abundant rainfall and mild temperatures—environmental conditions known to favour the growth of P. chartarum in pasture.
The aim of this study was to characterise the clinical, imaging, pathological and histopathological findings associated with this outbreak and to investigate a potential link with chronic sporidesmin exposure. The results support the existence of an atypical clinical presentation of pseudopithomycotoxicosis involving the rostral nasal cavity that has not been previously described in sheep.
CLINICAL, IMAGING AND PATHOLOGICAL FINDINGS
A complete clinical examination was conducted individually on each of the six animals. The six adult ewes (4-7 years old) showed upper respiratory signs but maintained a generally good overall appearance (Figure 1). Body condition score was low (1.5-2/5). Temperature, respiratory rate, heart rate and mucous membrane colouration were within normal limits.
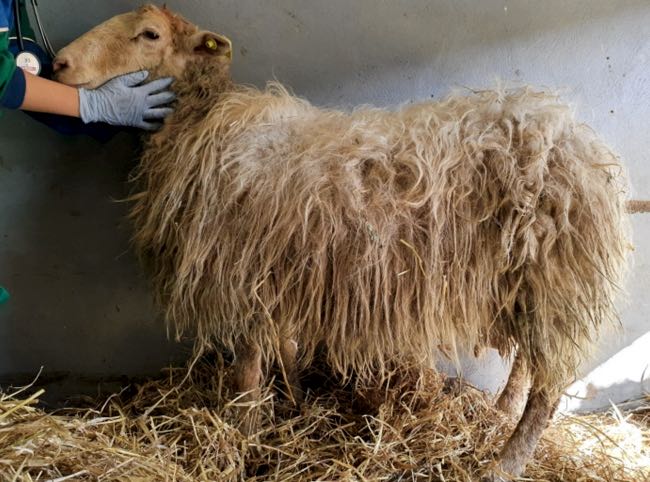
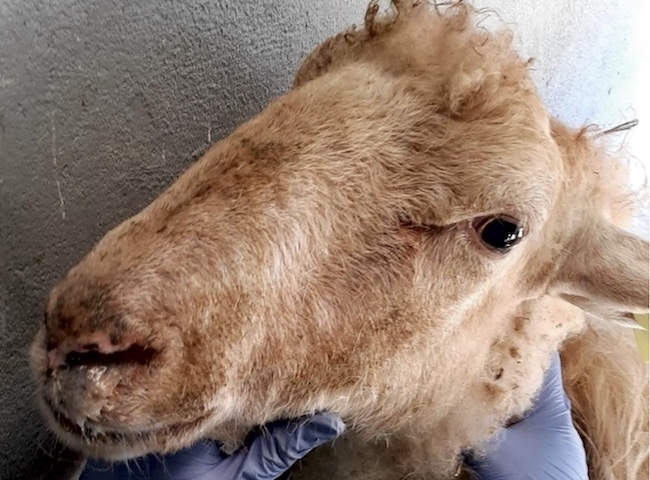
Figure 1. (2 images) Blond-faced Latxa ewes referred to SCRUM from the affected herds in the Basque country
The main abnormalities were respiratory. Bilateral serous nasal discharge was observed, together with continuous inspiratory dyspnoea and intense snoring, consistent with upper airway obstruction. Close examination of the nostrils revealed an evident reduction in nostril diameter due to partial occlusion of the nasal passages, markedly limiting airflow. Severity varied between animals and between sides (Figure 2). No other clinically relevant alterations were detected.
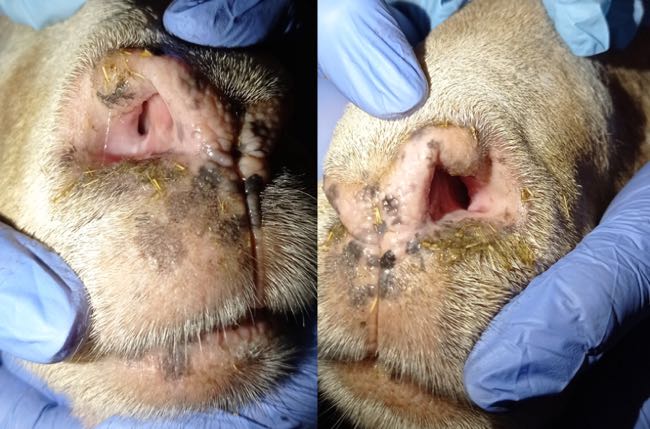
Figure 2. Macroscopic aspect of the nostrils of an affected ewe. This animal was severely affected by a marked inspiratory dyspnea accompanied by snoring. The right nostril was narrower than the left, although both sides were clearly occluded.
Whole blood and serum samples were collected for haematology and biochemistry. Haematology was within expected limits. In contrast, serum biochemistry showed consistent abnormalities in all animals, including increased gamma-glutamyl transferase (GGT) and alanine aminotransferase (ALT) and increased pancreatic lipase.
Given the clinical findings and prior experience with upper airway disease, ancillary tests were performed. Thermography did not identify consistent focal increases in temperature across the six animals, making common differentials such as chronic proliferative rhinitis (CPR) or enzootic nasal adenocarcinoma (ENA) less likely. Oestrosis had also been ruled out, as previous treatments targeting this condition failed to result in clinical improvement in the affected animals. Computed tomography (CT) of the head revealed increased soft tissue in the rostral nasal cavity, producing variable obstruction of the nasal meatuses in all sheep. On transverse CT images, soft-tissue thickening causing luminal narrowing was confined to the most rostral nasal cavity and decreased progressively on more caudal slices, where the nasal airway regained a wider lumen (Figure 3). This localisation of obstruction at the alar fold level explained the inspiratory dyspnoea and snoring.
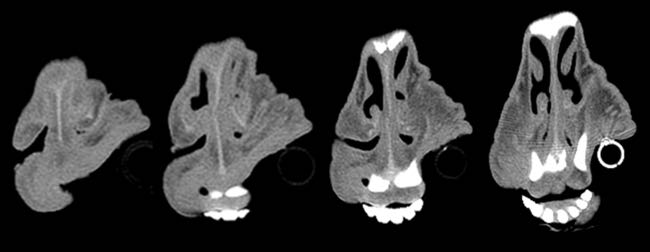
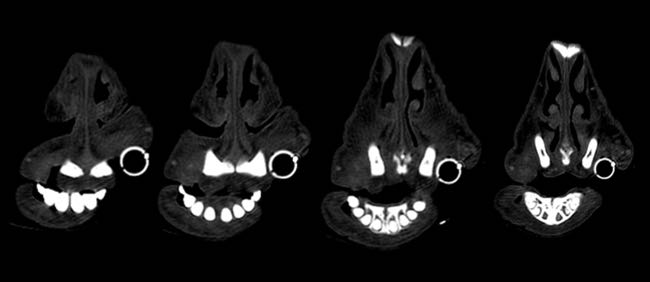
Figure 3. (2 images) Serial images obtained from the computed tomography scanner with different filters. Four sequential cuts of two sheep, from rostral (left) to caudal (right), show increased cellularity and almost complete blockage of the airway in the rostral area, which diminished as the images progress caudally.
The animals were then humanely euthanised for detailed post-mortem examination of the nasal cavity, liver and pancreas, as well as selected skin sites. Grossly, all sheep showed marked narrowing of the rostral nasal cavity due to proliferation of whitish tissue in the alar folds. The rostral segment of the dorsal nasal turbinate, alar fold, basal fold and adjacent areas of the ventral nasal turbinate were affected with variable caudal extension. The nasal mucosa in these areas was pearl white, slightly raised and patchily roughened, with the most severe lesions at the alar folds, resulting in a marked reduction of airflow (Figure 4).
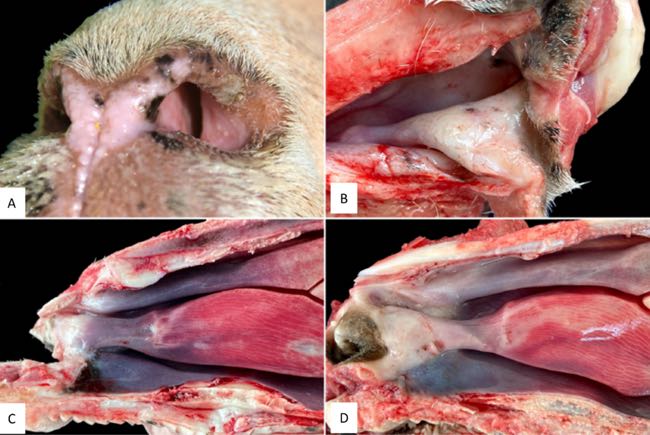
Figure 4. A. Left nostril of sheep, showing the narrow airway tract due to the presence of a whitish tissue. B. Dorsolateral view of the right nasal cavity. The alar fold and adjacent areas of the nasal mucosa appear thickened with a pearly white colour and have rough surfaces. C. Sagittal section. Gross changes were circumscribed to a small area on the alar fold, with a whitish pearl colour. D. Sagittal section. In this sheep, gross changes were more extensive, affecting from the alar fold to the dorsal, ventral nasal conchae, and ventral fold, all of them with a whitish-pearl colour.
Gross hepatic lesions were present in five of six animals. Two sheep showed atrophy and fibrosis of the left lobe. Three animals had marked liver atrophy with a firm, smooth, kidney-shaped appearance, consistent with severe interstitial fibrosis. In these cases, the gallbladder was also reduced and fibrotic, appearing as a dorsally displaced cord (Figure 5).
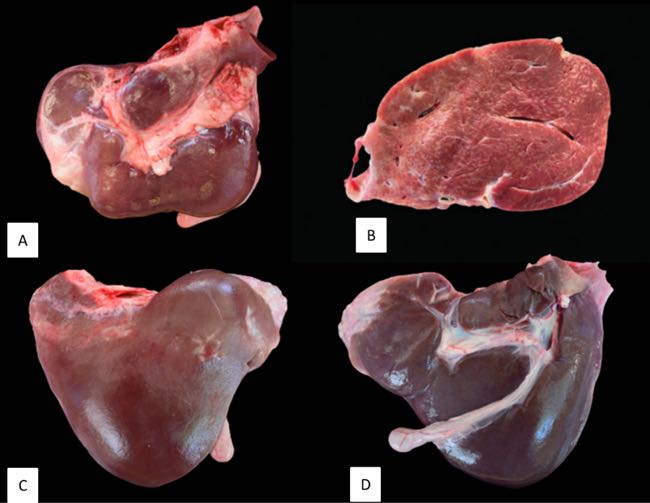
Figure 5. Hepatic gross lesions. A. Hepatic atrophy and fibrosis. B. Severely affected liver with interstitial fibrosis with band-like arrangement. C and D. Both sides of the same liver show a rounded surface, atrophy of the caudate lobe, increased size of the right and quadrate lobes and the presence of the gallbladder reduced and fibrosed, remaining only as a cord that has been displaced dorsally.
Microscopically, livers without evident gross lesions also showed periportal inflammation and fibrosis. Overall, the livers exhibited moderate-to-severe portal and bridging fibrosis, bile duct hyperplasia and scattered foci of hepatocellular necrosis. Vascular lesions were evident in vessels accompanying interlobular bile ducts, with severity paralleling the gross hepatic changes. Intimal proliferation with fibrosis and increased elastic fibres affected arteries and veins, mainly in interlobular areas, with fragmented elastic fibres admixed with collagen and mild mononuclear infiltration. In the gallbladder, lesions consisted of patchy epithelial necrosis/denudation, multifocal mucous gland proliferation, and fibrosis of the muscular and adventitial layers. No vascular lesions comparable to those in the liver were identified (De las Heras et al., 2022).
In the nasal cavity, lesions predominated in small arteries close to the epithelial surface. Many vessels showed eccentric intimal proliferation with variable oedema and fibrosis of the surrounding mucosa, without evidence of inflammation or calcification. Immunohistochemistry confirmed degenerative arterial changes, particularly in arteries within the alar folds and oriented towards the epithelial surface. Sheep with more severe hepatic lesions showed a higher proportion of affected nasal arteries (Figure 6).
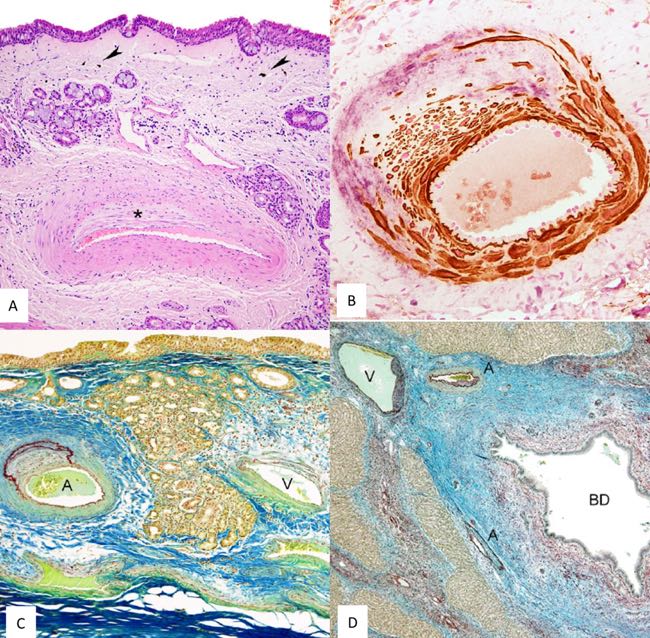
Figure 6. Microscopic lesions and immunohistochemistry exam. A. Histopathology of the nasal mucosa. Small artery located close to the surface of the nasal mucosa. Nasal epithelium remains unaltered. Intima of the artery shows eccentric proliferation (asterisk). Edema and fibrosis together with small groups of mononuclear cells, few neutrophils and melanotic cells are present (arrowheads). Hematoxylin-eosin. B. Immunohistochemistry of the nasal mucosa arteries. Small artery with intense intimal proliferation of smooth muscle cells and collagen III. Smooth muscle cells are shown with brown labeling and collagen III with light purple. Double immunohistochemistry smooth muscle actin and collagen III. C. Histopathology of the arteries of the nasal mucosa. Nasal mucosa showing one artery with intense intimal proliferation together with herniation and duplication of internal elastic membrane (A). Vein showing minimal changes in the wall (V). Orcein-Cajal's picrocarmin combination D. Histopathology of the liver. Interlobular septum showing 1 vein (V), 2 arteries (A) and 1 biliary duct (BD). Arteries and vein show sclerosis and intense fibrosis with proliferation of collagen and elastic fibers of interlobular septa. Intima vascular degenerative lesions are orientated to damaged BD. There are few mononuclear cells, mostly surrounding dilated biliary duct. Orcein-Cajal's picrocarmin combination (De las Heras et al., 2022).
Because facial eczema and nasal disease had been previously reported in the region, pasture sampling was performed to investigate potential mycotoxin exposure. Approximately 500-700 grams of fresh grass were collected from six grazing locations. Plant fragments were surface-sterilised in 75% ethanol, cultured on streptomycin-supplemented medium, and incubated at 26°C in darkness for 3-5 days, yielding Pseudopithomyces chartarum isolates. Conidial counts were estimated by washing samples with distilled water and quantifying conidia by light microscopy, with an average of 10² conidia/g of plant material.
The vascular lesions in hepatic and nasal arterioles were closely comparable, supporting a common aetiology. The distinct anatomical distribution may reflect a different exposure route to sporidesmin, with ingestion leading to portal uptake and hepatic injury, whereas inhalation or direct mucosal contact could lead to the rostral nasal presentation.
DISCUSSION
Facial eczema (FE), or pseudopithomycotoxicosis, is a well-recognised disease associated with the ingestion of pasture contaminated with P. chartarum spores containing the hepatotoxic mycotoxin sporidesmin. It is classically described as a seasonal hepatogenous photosensitisation affecting sheep and other grazing ruminants under conditions that favour fungal sporulation, particularly warm and humid climates. P. chartarum is a polyphagous saprophyte commonly found both in decaying plant material and as an endophyte within grasses (Dingley, 1962). Certain strains produce sporidesmin, an epidithiodioxopiperazine compound that induces liver injury, bile duct damage and obstruction, leading to phylloerythrin retention and, following sunlight exposure, photodermatitis of unpigmented skin areas, hence the term "facial eczema" (Gardiner et al., 2005; Di Menna et al., 2009; Riet-Correa et al., 2013).
Sporidesmin exerts a direct toxic effect, with its metabolites being less active or inactive (Jordan, 2020). Lesions are characteristically concentrated in bile ducts and the gallbladder, where the toxin induces epithelial degeneration, fibrosis and marked vascular changes. Vascular lesions associated with chronic exposure are typically located in vessels adjacent to damaged bile ducts and are oriented towards them (Jordan, 2020). Progressive hepatic atrophy, fibrosis, biliary hyperplasia and, in severe cases, cirrhosis characterise the chronic form of FE (Done et al., 1960; Mortimer, 1963; Fernández et al., 2021; Munday et al., 2021).
In the present study, the hepatic and gallbladder lesions, together with elevated serum GGT activity, were entirely consistent with chronic pseudopithomycotoxicosis, confirming exposure to sporidesmin. Importantly, the predominant and clinically most striking finding was not dermatological but respiratory, caused by a marked obstruction of the rostral nasal cavity. Computed tomography, necropsy and histopathology consistently localised the lesions to the alar folds and adjacent structures, where small arteries showed degenerative changes fulfilling criteria for arteriosclerosis. Notably, these vascular lesions closely resembled those observed in hepatic arterioles, supporting a shared pathogenic mechanism.
The presence of high concentrations of P. chartarum conidia in pasture samples further supports the diagnosis and is in agreement with previous reports from the same geographic area (Natorp, 2005; Berriatua et al., 2003). Taken together, these findings suggest that sporidesmin may also exert a local toxic effect on the rostral nasal cavity, likely related to direct contact during grazing. While sporidesmin toxicity has been extensively studied following ingestion, its potential impact on nasal tissues following inhalation or close mucosal exposure has not been previously documented.
A key aspect of this syndrome is that comparable obstructive nasal lesions have not been described in other regions where facial eczema is endemic, nor in other areas of northern Spain with similar climatic conditions (Di Menna et al., 2009; Riet-Correa et al., 2013; Fernández et al., 2021). This finding suggests that additional factors may contribute to the development of this presentation, including local climate, topography, very high spore loads—particularly in autumn—short pasture height, high sporidesmin concentrations, breed susceptibility and specific grazing or management practices. Under such conditions, the proximity of the nares to contaminated pasture may facilitate repeated exposure of the rostral nasal cavity to sporidesmin, leading to preferential damage of the alar folds and adjacent areas.
Finally, the apparent absence of reports of this presentation elsewhere may also reflect underdiagnosis. The condition may be overlooked by farmers and practitioners, particularly given its limited impact on short- and medium-term production parameters, despite its high prevalence and striking clinical signs. Increased awareness of this respiratory manifestation may therefore expand the recognised clinical spectrum of FE in sheep.
CONCLUSION
Obstructive rhinopathy associated with sporidesmin-producing P. chartarum represents a poorly recognised and understudied clinical presentation characterised by obstruction of the nasal passages at the level of the alar cartilage. Although farmers did not report an evident reduction in productivity, the marked respiratory compromise and the severe chronic hepatic lesions observed indicate a substantial negative impact on animal welfare and potentially on long-term performance. Further studies are required to elucidate why this presentation appears to be restricted to specific breeds and geographic areas, whereas elsewhere this mycotoxicosis is classically associated only with ovine FE.
REFERENCES
- Berriatua E, García-SanMartin MJ, Gómez N, et al. (2003) Ovine chronic nasal stertor (OCNAS): epidemiological and clinic-pathological description and relationship with sporidesmosis (ovine facial eczema) Proceeding of the 10th international symposium on veterinary epidemiology and economics Published 2003. Accessed March 14, 2024 www.sciquest.org.nz
- De las Heras M, Lacasta D, Reséndiz RA, Rivas A, Garzianda A, de Miguel R, Ruiz H, Castells E, González V and Ferrer LM (2022) Chronic pithomycotoxicosis associated with obstructive rhinopathy in sheepVeterinary Pathology 59(6) pp 950-959
- Di Menna ME, Smith BL and Miles CO (2009) A history of facial eczema (pithomycotoxicosis) research. New Zealand Journal of Agricultural Research 52 pp 345-376
- Dingley JM (1962) Pithomyces chartarum, its occurrence, morphology and taxonomy New Zealand Journal of Agricultural Research 5 pp 49-61
- Done J, Mortimer PH and Taylor A (1960) Some observations on field cases of facial eczema: liver pathology and determination of serum bilirubin, cholesterol, transaminase and alkaline phosphatase Research in Veterinary Science 1 pp 76-83
- Fernández M, Pérez V, Fuertes M, et al. (2021) Pathological study of facial eczema (Pithomycotoxicosis) in sheep Animals 11 p 1070
- Gardiner DM, Waring P and Howlett BJ (2005) The epipolydithiodioxopiperazine (ETP) class of fungal toxins: distribution, mode of action, function and biosynthesis Microbiology 151 pp 1021-1032
- Jordan TW (2020) The cellular and molecular toxicity of sporidesmin. New Zealand Veterinary Journal 68 pp 203-213
- Mortimer PH (1963) The experimental intoxication of sheep with sporidesmin, a metabolic product of Pithomyces chartarum. IV. Histological and histochemical examinations of orally-dosed sheep Research in Veterinary Science 4 pp 166-185
- Munday JS, Ridler A, Aberdein D, et al. (2021) Chronic facial eczema in sheep: description of gross and histological changes in the liver and association with serum gamma-glutamyltransferase activity at the time of sporidesmin intoxication New Zealand Veterinary Journal 69 pp 104-112
- Natorp JC (2005) Conduite à tenir lors d'oblitération nasale ovine Point Veterinaire 256 pp 40-44
- Riet-Correa F, Rivero R, Odriozola E, et al. (2013) Mycotoxicoses of ruminants and horses Journal of Veterinary Diagnostic Investigation 25 pp 692-708