
CASE NOTES
Managing Border Disease Virus in Commercial Flocks
Evie Duggan, District Veterinarian Cowra
Posted Flock and Herd August 2025
Reproductive Investigations
It is notoriously difficult to make a conclusive diagnosis in reproductive investigations following poor scanning or lamb marking rates. Abortion investigations are often just as challenging even though the point of loss has been identified. In their review of ovine abortion and stillbirth investigations, Clune & colleagues (2021) found that an infectious aetiology was diagnosed in 46% of the 57% of cases that reached a diagnosis reported by the pathologist.
If an investigation is instigated based on marking or weaning rates, it is important to remember that typical perinatal mortalities range from 10-35% (Shephard et al., 2022), with a perinatal mortality across the Australian sheep industry of 20-30% of lambs born dying before weaning and 74% within 3 days of birth (Jacobson et al., 2020). A problem should be defined on historical records from a property and what they consider as 'normal'.
It is more likely that an infectious agent will be diagnosed if more samples are submitted (Clune et al., 2021). Submitting veterinarians should not hesitate to submit autolysed tissues. While culture and histopathology may be negatively affected, molecular techniques and culture for the isolation of infective agents is still worthwhile. Samples to submit include:
- Pericardial fluid
- Fresh & fixed lung, liver, kidney, heart, spleen, brain & placenta
- Abomasal fluid.
(Kirkland, Hazelton & Duggan, 2021)
Inclusion of placental tissue increases the probability of making an aetiological diagnosis by 2.3 times. Requesting producers to collect any aborted material or perinatal dead lambs and placenta and to store these between 4-10°C before submission to the veterinarian, increases the likelihood of an infectious diagnosis being made (Clune et al., 2021).
The importance of submitting multiple foetuses / perinatal lambs cannot be underestimated in allowing a pattern to be discerned and preventing an incidental finding in one lamb being applied to the mob.
Serology of ewes often forms the basis for the investigation, often including but not limited to: Campylobacteriosis, Border Disease Virus, Toxoplasmosis, Listeriosis and Q Fever. Unless titres are paired with titres timed closely to when foetal losses occurred, it is difficult to interpret their relevance (Clune et al., 2021). It is suggested that bloods are submitted from 10 aborted ewes and 10 pregnant/unaffected ewes per management group (Kirkland, 2021).
Border Disease Virus positive in serology - now what?
Introduction
Border Disease Virus (BDV) is a Pestivirus within the same genus as the commonly encountered Bovine Viral Diarrhoea Virus (BVDV). BDV can infect hosts either transiently, which is mostly inapparent in sheep, or persistently (Schweizer & Peterhans, 2014). Similarly to BVDV, BDV causes reproductive impacts in flocks following infection of the foetus if naïve sheep are exposed for the first time during pregnancy. Depending on the stage of gestation, these impacts range from embryonic loss/abortion, the birth of weak, ill-thrifty lambs with a higher than usual perinatal mortality rate to the birth of persistently infected (PI) lambs (Figure 1).
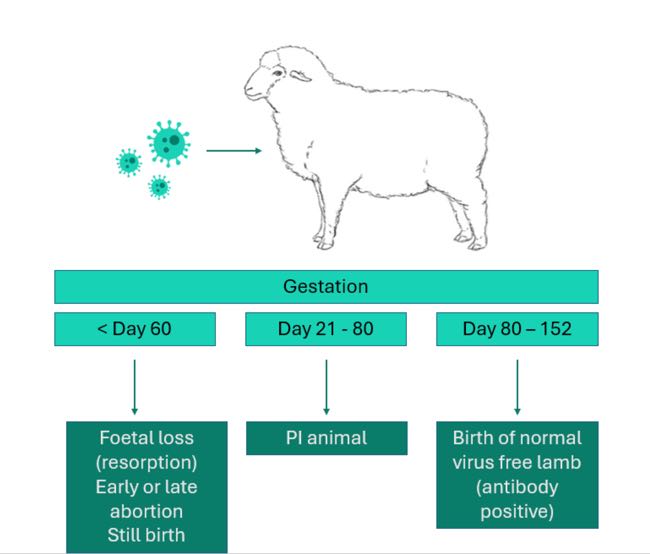
Figure 1. Graphic showing the consequence BDV infection depending on stage of gestation
PI lambs are born antibody negative but virus positive, shedding virus their entire lives and providing a reservoir of disease in a flock (Prell et al., 2023). Recent work has reported some PI lambs surviving for over 12 months which is longer than has been previously assumed in commercial flocks (Parrish et al., 2025). These PI animals are the primary source of infection, consistently shedding the virus in body secretions with transmission typically occurring through nose-to-nose or sexual contact. Acutely infected animals, aerosolised virus and contaminated fomites can also be sources of infection (Prell et al., 2024).
Management in a commercial flock
In regards to practical on-farm management, the most important thing to remember is that many of the principles of management of BVDV in cattle are quite different to the principles of management of BVD in sheep.
Expected losses
Typically outbreaks of BDV cause varying rates of abortion and increased perinatal mortality in the first year as the virus spreads through a naïve flock, but in following years losses are insignificant as the adult ewes develop immunity (Newcomer et al., 2020). Introduced ewes will obviously be at risk, if naïve. In the author's experience,outbreaks and high titres are more commonly seen in maidens that are joined as lambs, and/or spend a period of time in confinement.
PI's
In one case of BDV, lambs were tested at marking. Lambs with abnormal fleece were classified as H1 (extra hair around face & legs) or H2 (obviously hairy coat) and targeted for PACE testing. A high proportion of lambs with abnormal fleeces were confirmed as PI, while nearly all H2 were infected (98.9%) (Kirkland et al., 2021).

Figure 2. PI lamb (on right) in comparison to normal lamb at marking

Figure 3. PI lamb (fore) with abnormal fleece (H2) compared to 'normal' lambs
These findings suggest that in a flock with known exposure of the ewes to BDV, that identification of 'H2' PI's would be a cost-effective solution to the cost prohibitive PACE testing (minimum $11.65/hd) of large numbers of animals. H2's could be removed from the main population, reducing the PI's in the flock and therefore reducing the viraemic load.
Survival of PI's
In a case where PI's were identified and followed within a commercial operation, 75% of known PI's had died by weaning (5 months of age), while 15% of PI's survived for at least 12 months (Parrish et al., 2025).
Is it worth identifying the PI's to use as vaccinators?
A study conducted by Prell and colleagues (2023) showed insufficient seroconversion in mixed age ewes during exposure to PI lambs during a 2-15 day exposure period, at a generous ratio of 1 PI : 25 ewes.
Based on the above results, Prell & colleagues (2023) recommended a much longer exposure period or the use of much larger numbers of PI's (the latter unlikely to be attainable given their survival rates).
A containment feed lot may provide the opportunity to expose ewes to PIs at a ratio of of ≥1:25, with the proviso that they are removed 4-11 days prior to joining (Kirkland, 2024). However, this can be logistically challenging as it requires:
- identifying the PI's at lamb marking (minimum $11.65/hd if PACE testing, excluding time & labour, alternatively class in 'H2's' visually as suspect PI's),
- enough PI's available and
- an extended period of time in containment prior to joining.
If the use of PI's to gain seroconversion was to be pursued, it is recommended by Prell and colleagues (2023) to compare virus concentration in the surviving PI's. It is thought that there may be variability in viraemic load and therefore the volume of virus individual PI's shed. In practice however, it is assumed that all PI's available would be utilised.
The price for grain may make containment feeding, purely for the purpose of exposure to a PI, uneconomical.

Figure 4. PI lamb (on right) compared to normal lamb at weaning (5 months of age)
Is vaccination an option?
Currently there is no commercial vaccine available for BDV control. A highly effective BDV vaccine has been developed (currently being used as a model for other vaccines), however it is not available at this time (P Kirkland, personal communication, June 2024).
It has been shown that the BVDV vaccine is not protective against BDV in sheep (Prell et al., 2023) (Meyer et al., 2021).
Is eradication an option?
Successful BVDV control programs in other countries have shown that removal of PI animals results in viral extinction in the host population (Schweizer & Peterhans, 2014). Testing of all lambs would not be feasible and cost prohibitive, and so eradication of BVD in Australian flocks is not a practical management option.
Biosecurity Implications
Within flock
If groups of ewes (typically age groups or lines of ewes bought in) are identified as being naïve, while there is evidence of seroconversion amongst the rest of the flock, it is suggested to isolate that group while they are pregnant to prevent exposure to a PI during this high-risk time.
Properties have successfully prevented outbreaks of BDV between lines of sheep by utilising an existing road as a boundary between the two operations (Kirkland et al., 2021) showing that an outbreak can be contained within a flock. This same flock showed marked difference in serology between the two breeds of sheep they ran (Merino and Composite) showing that simple segregation in mobs was enough to prevent widespread circulation of the virus in this flock (Parrish et al., 2025).
Between flocks
When purchasing in young ewes, the likelihood of a PI being present is higher compared to older ewes. If the producer's flock is naïve, serology to establish the vendor-bred flock status and therefore likelihood of a PI being present should be considered.
If BDV is present on farm and replacement ewes are purchased, thought should be given to their isolation during gestation to prevent any contact with potential PI's on farm.
For properties that have BDV circulating, if maiden ewe lambs are managed in isolation, in theory the flock would transition to a naïve flock as the presence of PI's is reduced and contact between PI's (presumably younger animals) and adult ewes is limited.
Implications for cattle
Pestiviruses can move between sheep and cattle. Braun & colleagues (2012 & 2019) note that inter-species infection appears to occur particularly from cattle to sheep, and less commonly from sheep to cattle. BDV was less efficient than BVDV at infecting cattle in Switzerland (Braun et al., 2012), however cattle persistently infected with BDV have only rarely been observed in the field and in the author's opinion would be significantly less likely to occur under Australian commercial conditions compared to those in Switzerland.
Comingling of sheep and cattle, particularly during lambing provides the highest risk for BDV infection (Braun et al., 2019). BDV has been deemed to not be of significant risk to BVDV eradication programs in regions such as Scandinavia, therefore it can be assumed that the risk of transmission from Australian sheep to cattle is low (Schweizer & Peterhans, 2014).
Conclusion
BDV is an interesting and complex disease. Unlike BVDV in cattle, BDV in sheep appears to transmit inefficiently. Environments such as containment pens / feedlots create an increased risk of transmission between PI's and naïve animals as stocking density and contact between animals increases. However, even in those conditions, Prell and colleague's (2023) work shows that many PI's would be required, to obtain protective seroconversion, with 1:25 not being successful. For most commercial flocks where BDV is diagnosed, it is unlikely to cause significant losses again as seroconversion and immunity is obtained in those animals exposed (Newcomer et al., 2020). Based on this and the expected life span of most PI's, it is this author's opinion that for commercial operations it is not worth investing the time and money in identifying PI's, unless ewes are joined as lambs. In that case, it is suggested that ewe lambs are classed at weaning to reduce the number of PI's circulating after joining in such an immunologically naïve group of pregnant animals.
References
- Braun U, Reichle SF, Reichert C, Hassig M & Stalder H (2014) Sheep persistently infected with Border disease readily transmit virus to calves seronegative to BVD virus Veterinary Microbiology 168:98-104 dx.doi.org
- Braun U, Hilbe M, Peterhans E & Schweizer M (2019) Border disease in cattle The Veterinary Journal 246:12-20 doi.org
- Clune T, Beetson S, Besier S, Knowles G, Paskin R, Rawlin G, Suter R & Jacobson C (2021) Ovine Abortion and Stillbirth Investigations in Australia Australian Veterinary Journal 99(3):72-78 doi.org
- Jacobson C, Bruce M, Kenyon P, Lockwood A, Miller D, Refshauge G & Masters G (2020) A Review of Maternal Dystocia Meat & Livestock Australia Limited mla.com.au
- Kirkland P (2024) Border Disease: In World Organisation for Animal Health's (13th Ed.) Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 6th pp 1-14
- Kirkland P, Hazelton M & Duggan E (2021) Border Disease - A Rare Infection of Sheep? Conference presentation, District Veterinarian Conference Broken Hill, NSW, Australia [Unpublished]
- Meyer G, Combes M, Teillaud A, Pouget C, Bethune M & Cassard H (2021) Vaccination of sheep with bovine viral diarrhea vaccines does not protect against fetal infection after challenge of pregnant ewes with border disease virus Vaccines 9:805-819 doi.org
- Newcomer B, Cebra C, Chamorro M, Reppert E, Cebra M & Edmondson M (2021) Diseases of the hematologic, immunologic, and lymphatic systems (multisystem diseases) In Editors Pugh D, Baird A, Edmondson M & Passler T (3rd ed.) Sheep, Goat & Cervid Medicine pp 405-437, Elsevier doi.org
- Parrish K, Spiers Z, Hazelton M, Walker K, Duggan E, Graham W, Finlaison D & Kirkland P (2025) Large scale reproductive loss in sheep due to Border Disease virus infection, New South Wales, Australia [manuscript submitted for publication]
- Prell MM, McGrath SR, Kirkland PD & Allworth MB (2024) An investigation into the transmission and control of pestivirus in sheep in Australia Australian Veterinary Journal 102(3):60-66 doi.org
- Schweizer M & Peterhans E (2014) Annual Review of Animal Biosciences 2:141-163 doi.org
- Shephard R, Ware J, Blomfield B & Niethe G (2022) Priority list of endemic diseases for the red meat industry - 2022 update Meat & Livestock Australia Limited mla.com.au