
CASE NOTES
Nardoo (Marsilea drummondii) Causing Polioencephalomalacia, Cardiomyopathy and Mortality in First-Cross Ewes in the Coonamble District
Jillian Kelly, District Veterinarian Local Land Services, Coonamble and Anne Jordan, Pathologist, Elizabeth Macarthur Agricultural Institute, Menangle
Posted Flock & Herd May 2022
Introduction
Marsilea drummondii (Nardoo) is a plant known to cause polioencephalomalacia in sheep, and staggers and heart failure in horses. Historically it has been recorded throughout north-west (NW) NSW, often causing large mortalities events. This case is the first documented in sheep in the Coonamble district, which also featured an unusual case presentation - post-mortem changes consistent with heart failure, in addition to clinical polioencephalomalacia (PEM).
CASE REPORT/SERIES
In early September 2021, an investigation was conducted into deaths and illness in a mob of 800 first-cross ewes grazing a Moby barley crop (Hordeum vulgare) in the Quambone district of NW NSW. Six ewes were observed dead in the paddock, with another four showing signs of blindness. Winter and spring rainfall had been above average and as a result the paddock had not entirely been farmed to barley. One end was swampy, fed by a dam overflow. Inspection found this end of the paddock to grow almost exclusively M. drummondii (Nardoo) and Ranunculus pumilio (Ferny buttercup) (Figure 1).

Figure 1: M. drummondii and Ranunculus pumilio in situ

Figure 2: M. drummondii close up. (Photo credit: Justine McNally)
The sheep had been introduced to the barley paddock three weeks prior to the deaths. The ewes were offered a lick supplement of 50% agricultural lime and 50% salt, were recently vaccinated with 6-in-1 and they had been drenched with Q-drench (abamectin 1.0 g/L, albendazole 25.0 g/L, closantel 37.5 g/L, levamisole 40.0 g/L; Jurox) prior to going onto the barley crop.
Two ill ewes were examined and the mob was observed in the yards. The first ewe examined was head pressing into the corner of the yards. She had pink mucous membranes, a body temperature of 38.5°C and she was condition score 3/5. She had keratoconjunctivitis and a corneal ulcer in the left eye, but menace response was absent in both eyes and mentation was depressed. Hence the ocular inflammation was thought to be an incidental finding and the cause of the neurological signs to be central.
The second ewe examined was hyperexcited and running into fences consistent with blindness. Both eyes were clinically normal with an absent menace response. She had an unusually high head carriage, her ears were positioned forward, and she was hypermetric in the front legs. She had pink mucous membranes, her temperature was 38.5°C and her condition score was 3/5.
Another 4-5 ewes in the mob were noticed to be hypoaesthetic and dull.
These clinically affected animals were treated with Vitamin B1 (thiamine) injection (thiamine hydrochloride 125 mg/mL, 10mg/kg IM, Ceva Animal Health), and all made a complete recovery over the next 24-48 hours.
One of the dead ewes was autopsied. The most notable gross pathology finding was 1.5L of pleural fluid, spilling out once the thoracic cavity was penetrated. There was also excessive pericardial fluid. There were splash haemorrhages in the subcutaneous tissue covering the thoracic cavity and the epicardium. The liver was dark brown and firm in consistency with a nutmeg pattern. The jejunum was thickened and inflamed. Rumen pH was 6, and a M. drummondii sporocarp (reproductive part of the plant) was identified in the rumen.
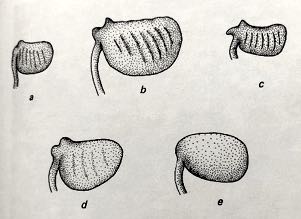
Figure 3: Marsilea sporocarps (with normal covering of hairs removed) a. M.augustifolia b. M.drummondii c. M.exarata d. M.hirsuta c. M.mutica (Cunningham, 1992)

Figure 4: Nardoo sporocarps in situ (Photo credit: Justine McNally)
Samples were sent to the NSW Department of Primary Industries (DPI) laboratory Elizabeth Macarthur Agricultural Institute (EMAI) and submitted under the Transmissible Spongiform Encephalopathy (TSE) program.
There were no CNS lesions suggestive of TSE in the sections examined. The most significant findings were in the heart, with extensive fibroplasia and small areas of acute necrosis, suggesting a repeated or ongoing cardiac insult. The observed cardiac histopathology was considered sufficient to cause significant cardiac compromise, consistent with the excess pericardial and thoracic fluid noted at autopsy. There were no changes suggestive of PEM in the sections of cerebrum examined and the brain did not fluoresce under a Wood's lamp. The liver sections examined showed vacuolar hepatopathy that was centrilobular and acute. There were no changes in the optic tract to support closantel toxicity, which could have accounted for the blindness.
Discussion
Clinical signs and deaths associated with PEM in sheep induced by grazing M. drummondii have been described in numerous cases throughout the years (Hurst, 1942; Eggleston 1975; Pritchard et al. 1978; McCleary et al. 1980, Slattery 1999, 2005).
In humans, the cardiac form of the disease is known as "wet beri-beri" and most famously led to the demise of the explorers Burke & Wills (Earl & McCleary, 1994). Thiamine is a water-soluble vitamin with a biologically active form known as thiamine pyrophosphate, which plays a critical role in carbohydrate metabolism and produces essential glucoses for energy by acting as a coenzyme. Thiamine deficiency renders pyruvate and some amino acids unavailable in many systems, with the cardiovascular system being particularly vulnerable (Lee et al. 2013).
In horses and pigs, the acute form of the disease has neurological and cardiac components that are believed to be due to thiamine deficiency (Jubb et al. 1992; Follis et al. 1943). However, many relevant case reports of M. drummondii in sheep that focus on PEM are historic, and information regarding cardiac lesions is sparse and incomplete. While Beckett (1984) described sheep that suddenly collapsed while mustering following ingestion of rock fern (Cheilanthes sieberi, a plant with similar toxic principles), this clinical presentation has not been described in sheep grazing M. drummondii. The cardiac syndrome has also not been observed in experimentally induced cases of M. drummondii intoxication in sheep (Watt & Bunker, 2011).
The clinical signs of PEM responded to thiamine injection, supporting a diagnosis of M. drummondii toxicity, despite the lack of histological changes consistent with PEM. The absence of histological changes is not uncommon in these cases (Shaun Slattery, pers. comm.). There was some artifactual vacuolation of the sections, which could mask mild genuine changes, and histological changes of PEM can also be multifocal. The brain did not fluoresce under the Wood's lamp, which suggests that severe necrosis is unlikely, however mild lesions could still be present. The animals were known to be grazing M. drummondii and the plant was found in the rumen. No further clinical signs were seen after the animals were shifted to another paddock without M. drummondii present.
The authors feel it is likely that thiaminases in M. drummondii caused both the brain and cardiac lesions and consider that the response to thiamine supports this hypothesis. We do recognise that the cardiac lesions are non-specific and can be seen with other toxic insults or Vitamin E/selenium deficiency. However, no other toxins were identified, the farm is not in a selenium deficient area, and vitamin E deficiency is unlikely in sheep with access to green feed.
It would be worthwhile to submit fixed cardiac tissue for histological examination in cases of suspected M. drummondii poisoning. If cardiac insults are found Vitamin E/Selenium deficiency needs to be excluded as a potential cause of the histological changes.
Acknowledgements
Thanks to Justine McNally & Shaun Slattery for sharing their experiences of M. drummondii toxicity in Western NSW.
References
- Cunningham GM, Mulham WE & Leigh JH. Plants of western NSW. CSIRO Publishing, 1992
- Earl, JW & McCleary BV. 1994. Mystery of the poisoned expedition. Nature, 368:683-684
- Eggleston GW. 1975. Nardoo poisoning in sheep. NSW Veterinary Proc. 11:29-30
- Jubb KVF, Kennedy PC & Palmer N. Pathology of Domestic Animals, 4th edition. P1658. 1992
- Follis RH, Miller MH, Wintrobe MM, Stein HJ. 1943. Development of Myocardial Necrosis and Absence of Nerve Degeneration in Thiamine Deficiency in Pigs. Am. J. Pathol.Mar;19(2):341-357
- Hurst, E. The poison plants of NSW. Snelling Printing Works Pty Ltd. 1942
- Lee HS, Lee SA, Shin HS, Choi HM, Kim SJ, Kim HK & Park YB. 2013. A case of cardiac beriberi: a forgotten but memorable disease. Korean Circ. J. 2013 Aug;43(8):569-72
- McCleary BV, Kennedy CA & Chick BF. 1980. Nardoo, bracken and rock ferns cause vitamin B1 deficiency in sheep. Agric. Gaz. NSW 91(5):40-43
- Pritchard D, Eggleston GW & Macadam JF. 1978. Nardoo fern and polioencephalomalacia. Australian Veterinary Journal 54:204-205
- Yeoman FT. 1946. Suspected Nardoo Poisoning. Institute of Inspectors of Stock of NSW Year Book www.flockandherd.net.au/archive
- Slattery S. 1999. Nardoo poisoning as a sporadic condition. Australian Sheep Veterinary Soc. Newsletter, pp4-5
- Slattery S. 2005. Unusual presentations of Chielanthes sieberi and Marsilea drummondii poisonings in the Narrabri RLPB district. www.flockandherd.net.au/archive
- Watt B & Bunker E. 2011. Fern intoxication in sheep and cattle. Poisonous plants workshop 2005. www.flockandherd.net.au